
If you’re studying chemistry, chances are you’ve come across the terms atoms, ions, and isotopes. These concepts are fundamental to understanding the building blocks of matter. In this article, we’ll provide an answer key for the atoms, ions, and isotopes chart, breaking down the essential information you need to know.
The chart includes essential details about each concept, such as the definitions, examples, and key characteristics. By using this answer key, you’ll be able to comprehend the differences between atoms, ions, and isotopes while also understanding how they relate to one another.
Understanding atoms is crucial to understanding the structure of matter. Atoms are the basic units of matter that make up everything around us. The chart will provide examples of various elements and their atomic structures, allowing you to visualize the composition of different atoms.
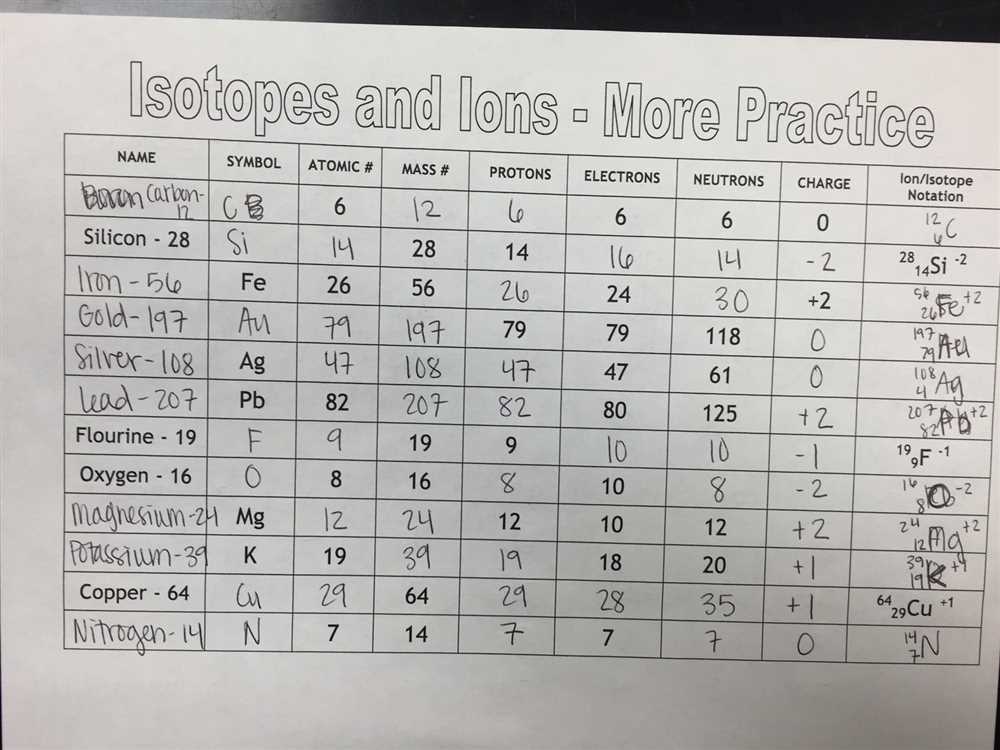
Ions, on the other hand, are atoms that have gained or lost electrons, resulting in a charge. The chart will explain the difference between cations and anions and provide examples to help you grasp the concept. Additionally, you’ll learn about the importance of ions in chemical reactions and their role in creating compounds.
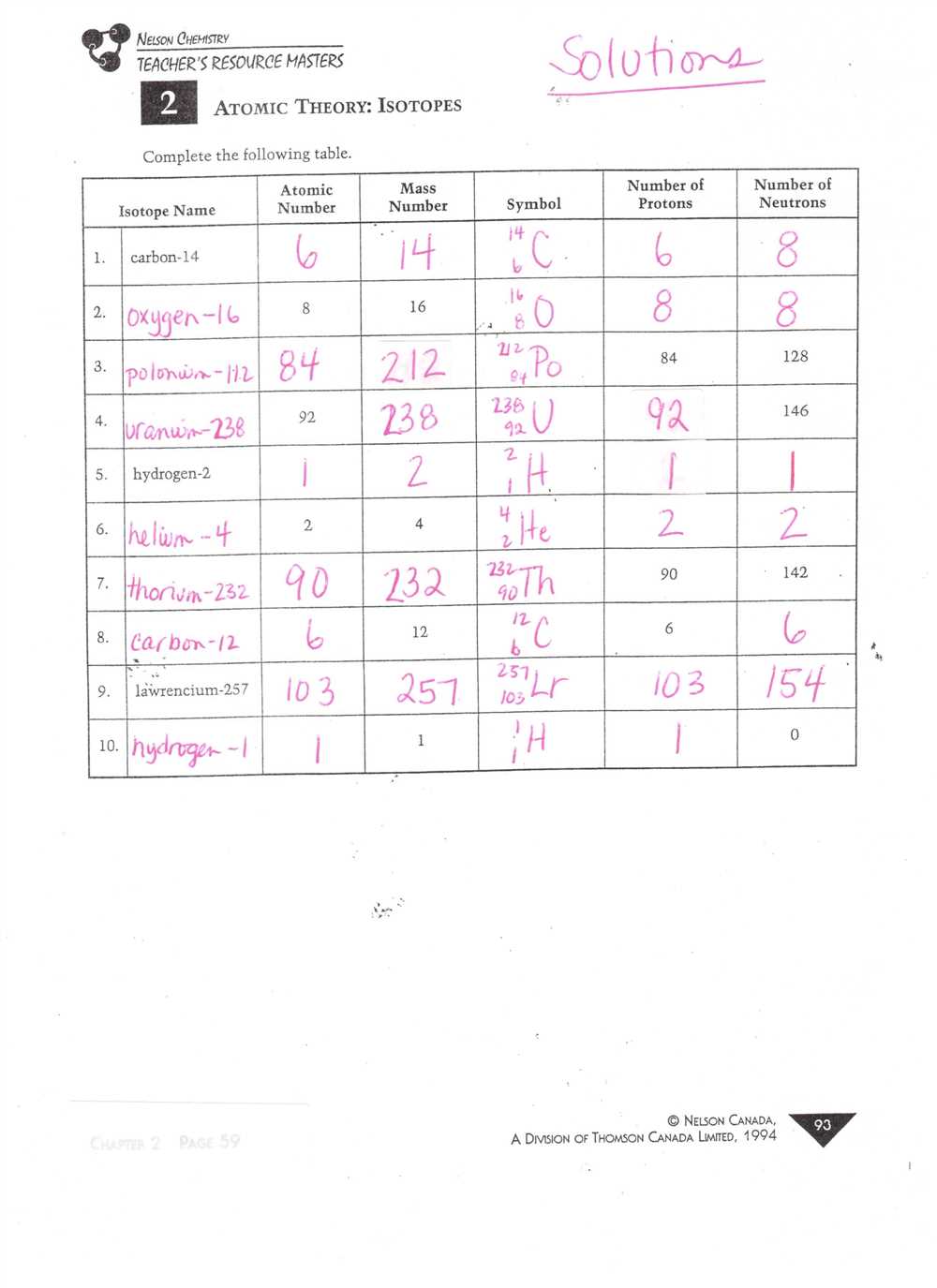
Isotopes are another important aspect of the chart. Isotopes are atoms of the same element that have different numbers of neutrons. This affects their atomic mass but not their chemical properties. The chart will provide examples of isotopes and help you understand how they can be used in various scientific applications, such as radiocarbon dating.
What are atoms, ions, and isotopes?

Atoms, ions, and isotopes are fundamental concepts in the field of chemistry that help us understand the building blocks of matter. Each of these terms represents different characteristics of an element and its constituent particles.
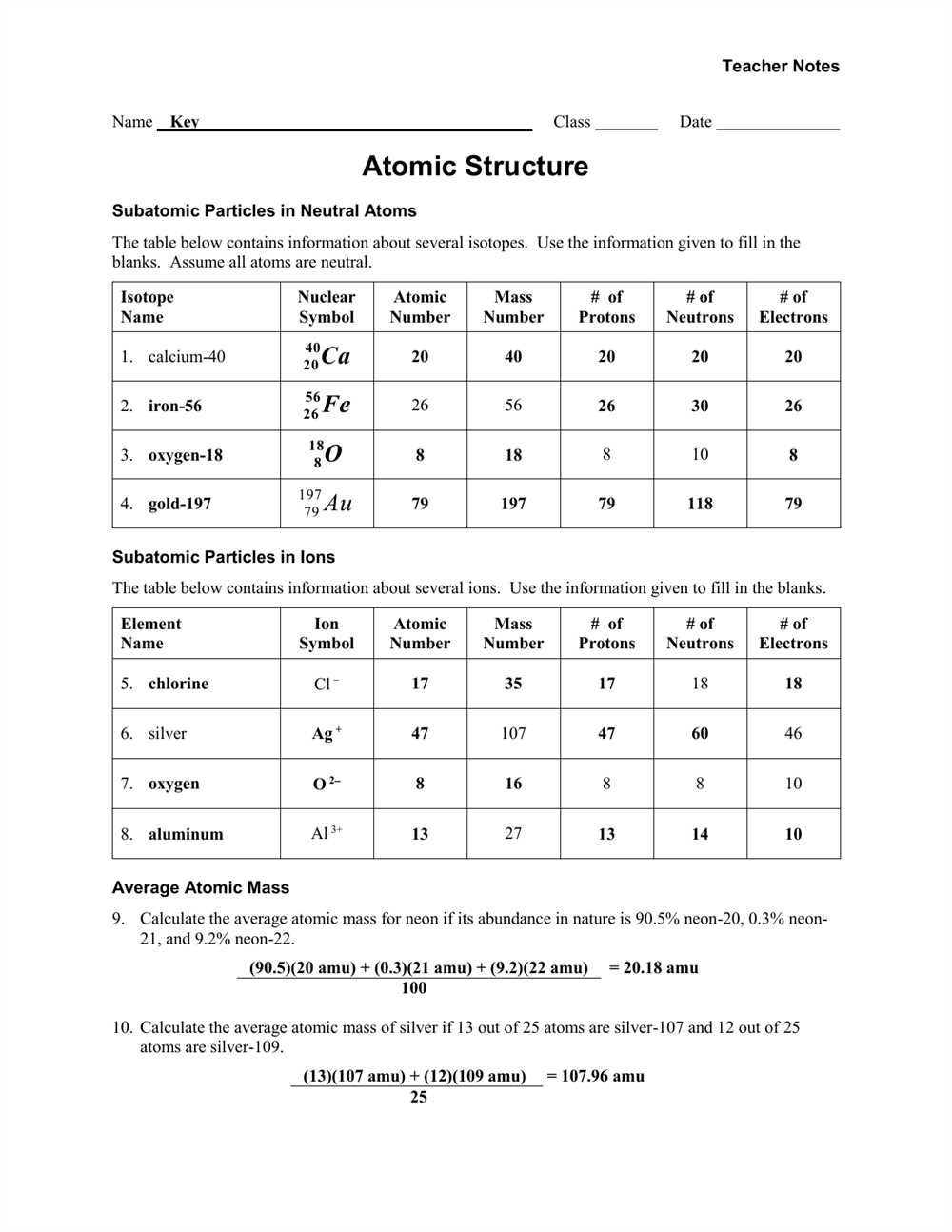
Atoms are the smallest units of matter that retain the properties of an element. They are made up of subatomic particles such as protons, neutrons, and electrons. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. The number of protons determines the element’s atomic number, which defines its unique identity.
Ions are charged particles that are formed when an atom gains or loses electrons. When an atom gains electrons, it becomes negatively charged and is called an anion. On the other hand, if an atom loses electrons, it becomes positively charged and is called a cation. Ions play a crucial role in chemical reactions and the formation of compounds.
Isotopes are atoms of the same element that have a different number of neutrons. This leads to variations in their atomic mass but does not affect their chemical properties significantly. Isotopes are identified by the element’s name along with their atomic mass number, which represents the total number of protons and neutrons in the nucleus of an atom.
In summary, atoms are the building blocks of matter, ions are charged particles resulting from the gain or loss of electrons, and isotopes are atoms of the same element with different numbers of neutrons. Understanding these concepts is crucial for understanding the behavior and properties of different elements and their interactions in chemical reactions.
Definition and Characteristics of Atoms
Atoms are the basic building blocks of matter. They are the smallest units of matter that retain the properties of an element. Atoms are made up of three types of particles: protons, neutrons, and electrons. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. The protons and neutrons are located in the nucleus, which is at the center of the atom, while the electrons orbit around the nucleus in various energy levels.
Characteristics of Atoms:
- Atomic Number: Each atom has a unique atomic number, which is equal to the number of protons in the nucleus. It determines the identity of the element.
- Atomic Mass: The atomic mass of an atom is the sum of the number of protons and neutrons in the nucleus.
- Isotopes: Isotopes are atoms of the same element that have different numbers of neutrons. They have the same atomic number but different atomic masses.
- Ions: Ions are atoms that have gained or lost electrons, resulting in a net charge. They can be positively charged (cations) or negatively charged (anions).
- Electron Configuration: The electron configuration refers to the arrangement of electrons in the energy levels around the nucleus. It determines the chemical properties of an atom.
Atoms are incredibly small, with diameters on the order of picometers. Despite their small size, atoms are highly important in understanding the structure and behavior of matter. They combine to form molecules and compounds, and their interactions govern the properties and reactions of substances. The study of atoms is fundamental to chemistry, physics, and other scientific disciplines.
Definition and characteristics of ions
Ions are electrically charged particles that are formed when atoms gain or lose electrons. An ion with a positive charge is called a cation, while an ion with a negative charge is called an anion. The charge of an ion is determined by the number of electrons gained or lost. Positively charged ions have more protons than electrons, while negatively charged ions have more electrons than protons.
When an atom loses electrons, it becomes positively charged because the number of protons in the nucleus is greater than the number of electrons. Conversely, when an atom gains electrons, it becomes negatively charged because the number of electrons is greater than the number of protons. The transfer of electrons between atoms creates a balance in the charges, resulting in the formation of ions.
Ions play a crucial role in chemical reactions and the formation of compounds. They are attracted to oppositely charged particles, leading to the formation of ionic compounds. These compounds are composed of ions held together by strong electrostatic forces of attraction. The charges of ions are represented by superscripts next to the atomic symbol in chemical formulas and equations.
Ions can exist in various forms and are classified based on their charge. Monoatomic ions are formed from a single atom and have a single charge, while polyatomic ions are composed of multiple atoms and have a overall charge. Common examples of ions include sodium ions (Na+), chloride ions (Cl-), and carbonate ions (CO32-).
In summary, ions are electrically charged particles that are formed when atoms gain or lose electrons. They can be positively or negatively charged, depending on the number of electrons gained or lost. Ions are crucial in chemical reactions and the formation of compounds, and their charges are represented in chemical formulas and equations.
Definition and Characteristics of Isotopes
Isotopes are variants of a particular chemical element with the same number of protons but different numbers of neutrons in the atomic nucleus. In simpler terms, isotopes are different forms of an element that have the same atomic number but different atomic masses due to the varying number of neutrons.
Isotopes are represented by the element’s atomic symbol followed by a hyphen and the mass number. For example, carbon-12 and carbon-14 are two isotopes of carbon. Carbon-12 has 6 protons and 6 neutrons, while carbon-14 has 6 protons and 8 neutrons.
There are several important characteristics of isotopes to consider:
- Atomic Mass: Isotopes have different atomic masses. The atomic mass of an element is the weighted average of the masses of all its naturally occurring isotopes.
- Stability: Isotopes can be stable or unstable, depending on the ratio of protons to neutrons in the nucleus. Some isotopes are radioactive and undergo radioactivity, while others are stable and do not decay.
- Radioactive Decay: Radioactive isotopes, also known as radioisotopes, undergo spontaneous decay over time. This decay process involves the emission of radiation, such as alpha particles, beta particles, or gamma rays.
- Applications: Isotopes have various applications in fields such as medicine, industry, and environmental monitoring. For example, radioactive isotopes can be used in medical imaging, nuclear power generation, and carbon dating.
Understanding isotopes is crucial for fields such as chemistry, physics, and geology, as they play a fundamental role in determining an element’s properties and behavior. The study of isotopes allows scientists to investigate the origins of elements, track chemical reactions, and analyze the composition of substances in different contexts.
Atomic structure and composition
The atomic structure refers to the organization of protons, neutrons, and electrons within an atom. At the center of the atom is the nucleus, which contains positively charged protons and neutrally charged neutrons. Electrons orbit the nucleus in specific energy levels or shells.
Protons are subatomic particles with a positive charge. They determine the identity of the element and contribute to the atomic mass. The number of protons in an atom is known as the atomic number and is denoted by the symbol “Z”.
Neutrons are subatomic particles with no charge. They provide stability to the atom by balancing the repulsive forces between protons. The combined number of protons and neutrons in an atom is known as the atomic mass or mass number and is denoted by the symbol “A”.
Electrons are negatively charged particles that occupy specific energy levels around the nucleus. They contribute to the atom’s size and chemical behavior. The electron configuration of an atom describes the arrangement of electrons in their respective energy levels.
The composition of an atom can be represented using an atomic symbol, where the atomic number is placed as a subscript to the left of the element symbol, and the atomic mass is placed as a superscript to the left of the element symbol. For example, the atomic symbol for carbon is written as 12C, indicating an atomic number of 6 and an atomic mass of 12.
The understanding of atomic structure and composition is fundamental to many areas of science, including chemistry and physics. It provides insights into the behavior and properties of different elements, the formation of chemical bonds, and the behavior of matter at the atomic and molecular level.
Electron, Proton, and Neutron Distribution
In an atom, electrons, protons, and neutrons are distributed in different regions. The distribution of these particles determines the properties and behavior of the atom.
Electron Distribution: Electrons are negatively charged particles that orbit around the nucleus. They are arranged in energy levels or shells. The first energy level can hold a maximum of 2 electrons, the second level can hold a maximum of 8 electrons, and the third level can hold a maximum of 18 electrons. The electrons in the outermost energy level are called valence electrons, and they determine the reactivity and chemical properties of the atom.
Proton Distribution: Protons are positively charged particles located in the nucleus of the atom. Each proton has a mass of 1 atomic mass unit (AMU). The number of protons in an atom is known as the atomic number and determines the identity of the element. For example, hydrogen has 1 proton, helium has 2 protons, and oxygen has 8 protons.
Neutron Distribution: Neutrons are neutral particles also located in the nucleus of the atom. Each neutron has a mass of approximately 1 AMU, just like a proton. The number of neutrons in an atom can vary, resulting in different isotopes of the same element. Isotopes have the same number of protons but different numbers of neutrons.
In summary, electrons orbit around the nucleus in energy levels, protons are located in the nucleus and determine the element’s identity, and neutrons also reside in the nucleus and contribute to the atom’s mass. The distribution of these particles determines the overall characteristics and behavior of the atom.
Relationship between atomic number and atomic mass
The atomic number and atomic mass are two fundamental properties of an atom that provide valuable information about its identity and characteristics. The atomic number is a unique identifier for each element and corresponds to the number of protons in the nucleus of an atom. It is represented by the symbol Z. The atomic mass, on the other hand, is the sum of the masses of protons, neutrons, and electrons in an atom. It is represented by the symbol A.
The relationship between atomic number and atomic mass can be understood by examining the structure of an atom. The atomic mass is primarily determined by the masses of protons and neutrons, as electrons contribute very little to the overall mass. Since the atomic number represents the number of protons, it is directly related to the atomic mass. In general, as the atomic number increases, so does the atomic mass.
To illustrate this relationship, let’s consider an example. Oxygen has an atomic number of 8, which means it has 8 protons in its nucleus. The atomic mass of oxygen is approximately 16 atomic mass units (amu). This value is obtained by adding the masses of 8 protons and 8 neutrons, which are the main contributors to the atomic mass. Therefore, we can conclude that as the atomic number increases from element to element, the atomic mass also increases.
It is important to note that there are exceptions to this relationship due to the presence of isotopes. Isotopes are atoms of the same element that have different numbers of neutrons. These variations in neutron numbers can affect the atomic mass while keeping the atomic number constant. Isotopes can be identified by their mass numbers, which indicate the total number of protons and neutrons in the nucleus. Understanding the relationship between atomic number and atomic mass is crucial in analyzing and studying the properties and behavior of different elements.
Role of Valence Electrons in Chemical Reactions
Valence electrons play a crucial role in chemical reactions as they are responsible for the formation and stability of chemical bonds. These electrons are found in the outermost energy level of an atom, also known as the valence shell. The number of valence electrons determines the chemical properties of an element, including its reactivity and ability to form compounds.
The valence electrons are involved in two types of chemical bonds: ionic bonds and covalent bonds. In ionic bonding, valence electrons are transferred from one atom to another, resulting in the formation of ions. Positive ions are formed when atoms lose valence electrons, while negative ions are formed when atoms gain valence electrons. The attraction between ions of opposite charges creates an ionic bond.
In covalent bonding, atoms share their valence electrons to achieve a stable electron configuration. This type of bonding occurs between nonmetallic elements. The shared electrons form a bond that holds the atoms together in a molecule. The number of covalent bonds that an atom can form is determined by the number of unpaired valence electrons it possesses.
The reactivity of an atom depends on the number of valence electrons it has. Elements with fewer valence electrons tend to lose or share electrons to achieve a stable electron configuration, making them more reactive. On the other hand, elements with nearly full or full valence shells are less reactive, as they have a stable electron configuration and do not readily form chemical bonds.
In conclusion, valence electrons are essential in chemical reactions because they determine an atom’s reactivity and ability to form bonds. Understanding the role of valence electrons allows chemists to predict the behavior and properties of elements and compounds, contributing to advancements in various fields of science and industry.