Collision theory is a fundamental concept in chemistry that helps explain the rate at which chemical reactions occur. Understanding the ins and outs of this theory is essential for students and researchers in the field.
The collision theory answer key provides a comprehensive guide to understanding the various factors that influence the rate of a chemical reaction. From the nature of the reactants to the temperature and concentration, each element plays a crucial role in determining the speed at which a reaction occurs.
This answer key breaks down the theory into easily understandable sections, covering topics such as effective collisions, activation energy, and the role of catalysts. By delving into the key principles of collision theory, students can gain a deeper understanding of how and why reactions happen.
Whether you’re studying for an exam or conducting research in the lab, the collision theory answer key is an invaluable resource. With clear explanations and illustrative examples, this guide will help you unlock the secrets behind chemical reactions and pave the way for future discoveries in the field of chemistry.
What is Collision Theory? A Comprehensive Answer Key
In chemistry, collision theory is an important concept that explains the rate of chemical reactions. It states that for a reaction to occur, the reactant molecules must collide with sufficient energy and in the correct orientation. This theory helps scientists understand the factors that influence reaction rates and allows them to predict and control the outcomes of chemical reactions.
The key components of collision theory include the collision frequency, the orientation factor, and the activation energy. Collision frequency refers to the number of collisions per unit time, and it is influenced by factors such as temperature, concentration, and pressure. The orientation factor measures the probability of reactant molecules colliding in the correct orientation to form products. Finally, the activation energy is the minimum energy required for a reaction to occur. It is important because only collisions with sufficient energy can overcome the activation energy barrier.
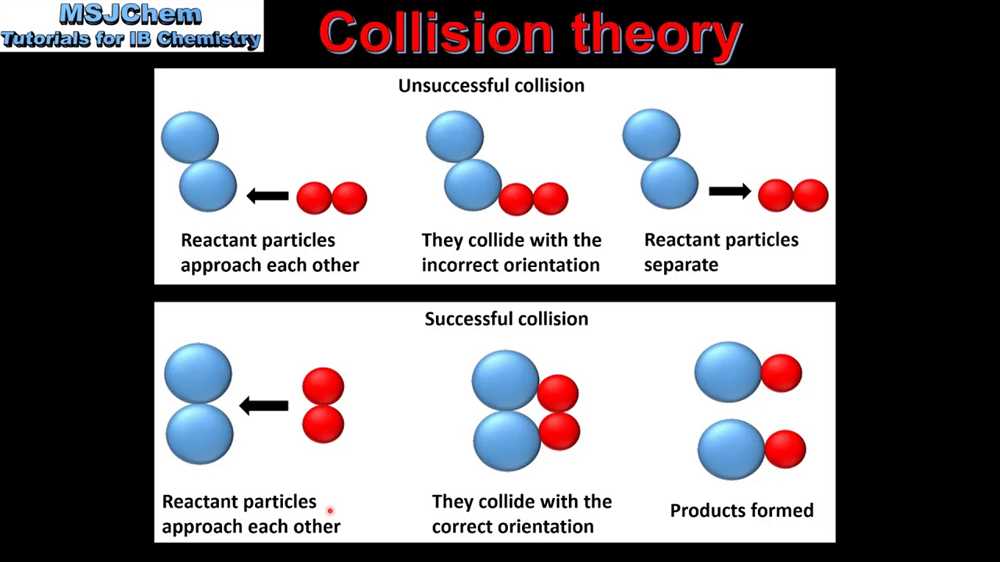
When a chemical reaction occurs, the reactant molecules must first collide. However, not all collisions result in a reaction. According to collision theory, only collisions that meet the criteria of sufficient energy and correct orientation can lead to a successful reaction. If the colliding molecules have insufficient energy, they will simply bounce off each other and no reaction will occur. Similarly, if the molecules collide with the wrong orientation, they will not be able to form the necessary bonds to produce the desired products.
Temperature plays a crucial role in collision theory. As temperature increases, the kinetic energy of the molecules also increases, leading to more frequent and energetic collisions. This results in a higher collision frequency and a greater probability of successful reactions. Concentration and pressure also affect collision frequency by influencing the number of molecules available for collisions. Higher concentrations and pressures increase the collision frequency, increasing the chances of successful reactions.
Collision theory has significant practical applications in various fields, including industrial chemistry. By understanding the factors that affect reaction rates, scientists and engineers can optimize reaction conditions to improve yields and efficiency. Additionally, collision theory provides insights into the mechanisms of complex reactions, allowing for the development of new catalysts and chemical processes. Overall, collision theory is a fundamental concept that helps us understand and control the world of chemical reactions.
Understanding the Basics of Collision Theory

In chemistry, the collision theory is an essential concept that explains the rate of chemical reactions. It states that for a reaction to occur, particles must collide with proper orientation and sufficient energy. The collision theory helps scientists understand how different factors, such as temperature and concentration, affect the rate of a reaction. By studying and analyzing collisions at the molecular level, scientists can gain insights into the mechanisms of chemical reactions.
A key aspect of the collision theory is the concept of collision frequency. Collision frequency refers to the number of collisions that occur per unit of time. It is influenced by factors such as the concentration of reactants, surface area, and temperature. For example, increasing the concentration of reactants or the surface area of the reactants will lead to a higher collision frequency, thereby increasing the reaction rate. Similarly, raising the temperature increases the kinetic energy of the particles, leading to more frequent and energetic collisions.
The collision theory also emphasizes that not all collisions result in a reaction. Only collisions that possess enough energy to surmount the activation energy barrier and the correct orientation of the particles can lead to a successful reaction. In other words, not all collisions are effective. This concept is crucial in understanding reaction mechanisms and designing experiments to optimize reaction rates.
To summarize, the collision theory is a fundamental concept in chemistry that explains how the rate of chemical reactions is influenced by factors such as temperature, concentration, and collision frequency. By understanding the basics of collision theory, scientists can gain a deeper understanding of reaction mechanisms and devise strategies to control and optimize reaction rates.
Key Principles of Collision Theory

Collision theory is a fundamental concept in chemistry that explains the rates at which chemical reactions occur. It is based on the idea that for a reaction to take place, reactant particles must collide with a certain amount of energy and with proper orientation. There are several key principles of collision theory that are essential to understanding chemical reactions.
1. Collision frequency: The rate at which reactant particles collide with each other is a crucial factor in determining the reaction rate. The more often particles collide, the greater the chance of a successful reaction taking place.
2. Energy barrier: In order for a collision to result in a reaction, the colliding particles must possess enough energy to overcome the activation energy barrier. This energy barrier represents the minimum energy required for a reaction to occur.
3. Proper orientation: In addition to energy, the particles must also collide with the correct orientation. This means that they must align in a way that allows the necessary molecular rearrangements to occur for the reaction to take place.
4. Effective collisions: Not all collisions between reactant particles result in a reaction. Only collisions that meet the criteria of energy and orientation are considered effective collisions. These collisions lead to the formation of products.
5. Reaction rate: The rate at which a reaction occurs is determined by the frequency of effective collisions. Increasing the concentration of reactants, temperature, or the presence of a catalyst can all increase the reaction rate by increasing the frequency of effective collisions.
Overall, collision theory provides a clear framework for understanding chemical reactions and the factors that influence their rates. By considering concepts such as collision frequency, energy barrier, proper orientation, and effective collisions, scientists can gain insights into the mechanisms and kinetics of various chemical reactions.
How Collision Theory Explains Reaction Rates
Collision theory is a fundamental concept in chemistry that explains how reaction rates are influenced by the collision of particles. According to this theory, for a chemical reaction to occur, particles must collide with sufficient energy and in the correct orientation. The rate of a reaction is dependent on the frequency of collisions and the fraction of collisions that have enough energy to overcome the activation energy barrier.
The role of collision energy: In order for a reaction to take place, particles must possess enough kinetic energy to overcome the repulsive forces between them and allow chemical bonds to be broken and formed. The higher the collision energy, the more likely it is for a reaction to occur. This is why increasing the temperature of a system generally increases the reaction rate, as it provides more energy to particles, increasing their collision energy and frequency.
The importance of collision orientation: Even if particles have sufficient energy, they may not react if their collision does not occur in the correct orientation. This is because chemical reactions involve atoms and molecules coming together in specific arrangements to form new bonds. If particles collide with the wrong orientation, they may simply bounce off each other without any reaction occurring. Hence, the probability of successful collisions is influenced by the spatial orientation of the particles involved.
Overall, collision theory provides an explanation for how the kinetics of chemical reactions are influenced by the collision of particles. By understanding the relationship between collision energy, collision orientation, and reaction rates, scientists can better predict and control the rates of chemical reactions, leading to advancements in fields such as drug development, materials science, and environmental chemistry.
Factors Affecting Collision Theory
The collision theory is a concept in chemistry that explains how chemical reactions occur through the collision of particles. According to this theory, for a chemical reaction to take place, the reacting particles must collide with sufficient energy and the correct orientation. However, several factors can influence the rate and effectiveness of these collisions.
1. Temperature: Temperature plays a significant role in the collision theory. Higher temperatures increase the kinetic energy of particles, making them move faster and collide more frequently. This leads to an increase in the rate of successful collisions and a higher reaction rate. Conversely, lower temperatures decrease the kinetic energy and collision frequency, resulting in a slower reaction rate.
2. Concentration: Concentration refers to the amount of reactant particles per unit volume. A higher concentration of reactants means there are more particles available to collide, increasing the chances of successful collisions. This leads to a higher reaction rate. On the other hand, lower concentrations result in fewer collisions and a slower reaction rate.
3. Catalysts: Catalysts are substances that increase the rate of a chemical reaction by providing an alternative reaction pathway with lower activation energy. They do not participate in the reaction itself but can significantly affect the collision theory. Catalysts provide a surface for reactant particles to adsorb, increasing the frequency of collisions and promoting the proper orientation. This lowers the activation energy required for the reaction to occur and increases the reaction rate.
4. Surface area: The surface area of a solid reactant can also impact the collision theory. Increasing the surface area by grinding or pulverizing a solid reactant exposes more particles to the surrounding reactants in the solution or gas phase. This increases the chances of successful collisions and enhances the reaction rate.
5. Pressure: Pressure is a factor that affects the collision theory in gas-phase reactions. Increasing the pressure of gas reactants increases the number of particles per unit volume. This leads to more frequent collisions and a higher reaction rate. Conversely, decreasing the pressure results in lower collision frequency and a slower reaction rate.
In conclusion, factors such as temperature, concentration, catalysts, surface area, and pressure can significantly affect the collision theory. By understanding and controlling these factors, chemists can manipulate the rate and effectiveness of chemical reactions.
The Role of Activation Energy in Collision Theory
In collision theory, activation energy plays a crucial role in determining whether a reaction will occur and at what rate. Activation energy is the minimum amount of energy required for a collision between reactant molecules to result in a successful reaction.
When reactant molecules collide, they must overcome a certain energy barrier known as the activation energy barrier in order to form the products of the reaction. This barrier is necessary because not all collisions between molecules lead to a reaction. Only those collisions that possess enough energy to overcome the activation energy barrier will result in a successful reaction.
The magnitude of activation energy determines the rate of the reaction. Reactions with a higher activation energy barrier will occur more slowly, as fewer collisions between molecules will possess enough energy to exceed the barrier. Conversely, reactions with a lower activation energy barrier will occur more quickly, as a larger proportion of collisions will result in a successful reaction.
Overall, activation energy acts as a kind of “threshold” that reactant molecules must surpass in order to produce a reaction. It serves as a critical factor in determining the feasibility and rate of chemical reactions, making it an essential concept in collision theory.
Collision Theory in Chemical Reactions: Examples and Applications
The collision theory is a fundamental concept in chemistry that explains how chemical reactions occur at the molecular level. According to this theory, for a reaction to take place, reacting particles must collide with sufficient energy and proper orientation. This concept helps scientists understand and predict the rate and outcome of chemical reactions in various systems.
An example that illustrates the collision theory is the reaction between hydrogen and oxygen to form water. In this reaction, hydrogen and oxygen molecules collide with each other and break their existing bonds. This collision provides the necessary energy for the formation of new bonds between hydrogen and oxygen atoms, resulting in the creation of water molecules. The collision theory helps explain why increasing the concentration of reactants or raising the temperature of the system increases the rate of this reaction. Higher concentrations or temperatures provide more frequent and energetic collisions between the molecules, increasing the likelihood of a successful reaction.
The collision theory has numerous applications in various fields of chemistry. One notable application is in the field of industrial catalysis. Catalysis involves the use of a catalyst to increase the rate of a chemical reaction. The collision theory helps scientists design and optimize catalysts by understanding the importance of surface area and proper orientation of reacting particles. By providing more surface area for collisions and ensuring proper alignment of reactants, catalysts can effectively increase the rate of reactions.
In conclusion, the collision theory is a fundamental concept in chemistry that explains how chemical reactions occur. It helps scientists understand and predict the rate and outcome of reactions by considering factors such as collision frequency, energy, and orientation. Through examples like the reaction between hydrogen and oxygen, and applications in areas such as industrial catalysis, the collision theory has proven to be a valuable tool in the field of chemistry.


