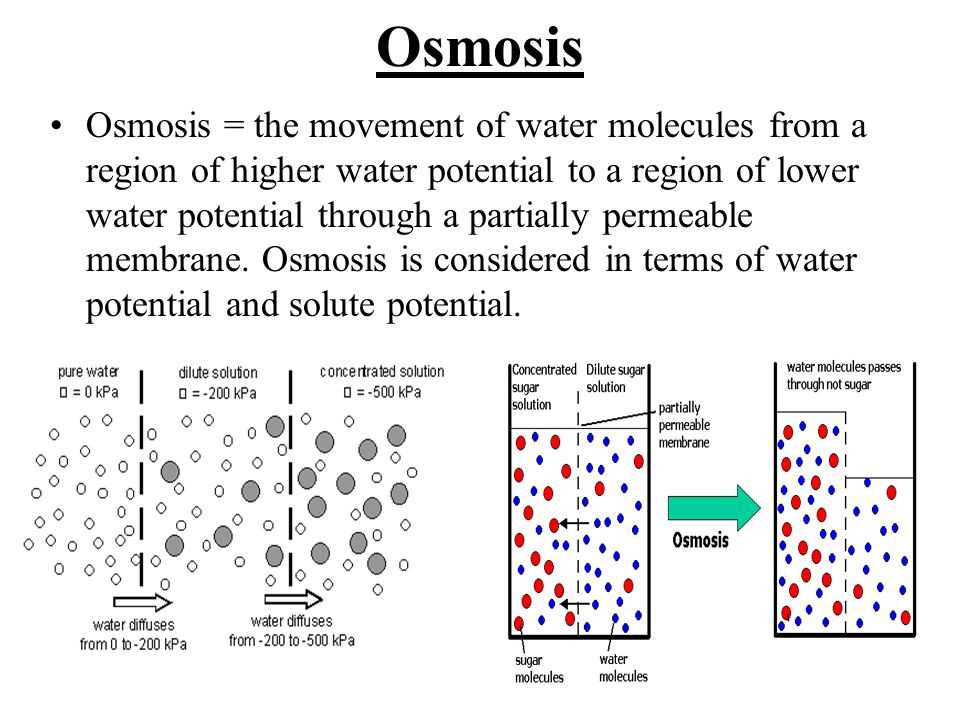
Understanding the concepts of diffusion and osmosis is crucial in the field of biology. These processes play a critical role in the transportation of molecules and the maintenance of cell homeostasis. Diffusion refers to the movement of molecules from an area of higher concentration to an area of lower concentration. On the other hand, osmosis is the movement of water molecules across a semi-permeable membrane from a region of low solute concentration to a region of high solute concentration.
Diffusion and osmosis problems are commonly encountered in biology and related fields. These problems often require an understanding of the key principles and equations associated with these processes in order to solve them. Having access to a comprehensive answer key can greatly assist students and professionals in evaluating and correcting their work.
The answer key for diffusion and osmosis problems provides definitive solutions and explanations for a variety of scenarios. It allows individuals to verify their understanding of the concepts and identify any mistakes made during problem-solving. Additionally, the answer key may offer alternative methods or approaches to solving the problems, enhancing the learner’s critical thinking skills.
By utilizing the diffusion and osmosis problems answer key, students and professionals can effectively assess their knowledge and master these essential biological processes. The key serves as a valuable resource for self-study, exam preparation, and classroom instruction.
Diffusion and Osmosis Problems Answer Key: A Comprehensive Guide
In the study of biology, understanding the concepts of diffusion and osmosis is crucial. These processes are fundamental to the movement of molecules and ions in biological systems. Diffusion refers to the passive movement of particles from an area of higher concentration to an area of lower concentration. Osmosis, on the other hand, is a specific type of diffusion that involves the movement of water molecules across a selectively permeable membrane.
To fully grasp the principles of diffusion and osmosis, it is important to be able to solve problems related to these concepts. Diffusion and osmosis problems often involve calculating the rate of diffusion or determining the direction of water movement in osmosis. They can be challenging, but with the right approach, they can be solved effectively.
One key to solving diffusion and osmosis problems is understanding the underlying concepts and principles. This includes knowing how concentration gradients affect diffusion rates and how osmosis is influenced by solute concentration and membrane permeability. By grasping these fundamental concepts, you can approach problems with a solid foundation.
Another important aspect of solving diffusion and osmosis problems is applying the appropriate formulas and equations. For example, Fick’s law of diffusion can be used to calculate the rate of diffusion, while the water potential equation can help determine the direction of water movement in osmosis. Familiarizing yourself with these formulas and understanding when to use them is key.
Additionally, it can be helpful to practice with a variety of diffusion and osmosis problems to develop your problem-solving skills. This can involve working through textbook exercises, participating in laboratory simulations, or seeking additional resources online. The more you practice, the more comfortable you will become with applying the concepts and formulas to different scenarios.
In conclusion, mastering diffusion and osmosis problems requires a comprehensive understanding of the concepts, familiarity with the relevant formulas, and ample practice. By honing these skills, you will be well-equipped to tackle any diffusion and osmosis problem that comes your way.
Understanding Diffusion and Osmosis: Key Concepts
Diffusion and osmosis are both fundamental processes that occur in biological systems, and understanding their key concepts is crucial for comprehending various biological phenomena. Diffusion refers to the spontaneous movement of molecules or particles from an area of higher concentration to an area of lower concentration, driven by the random thermal motion of particles. This process continues until equilibrium is reached, where the concentration is equal throughout the system.
On the other hand, osmosis is a specific type of diffusion that involves the movement of solvent molecules (usually water) across a selectively permeable membrane. The movement occurs from an area of lower solute concentration to an area of higher solute concentration, in order to equalize the concentration on both sides of the membrane. It is important to note that osmosis only applies to the movement of solvent molecules, not solute molecules.
In order to understand these processes more deeply, it is essential to grasp key concepts such as concentration gradient, membrane permeability, and isotonic, hypotonic, and hypertonic solutions. The concentration gradient refers to the difference in concentration of a substance between two areas, which drives the diffusion or osmosis process. The membrane permeability determines which molecules or ions can pass through the membrane, with some membranes allowing certain molecules to pass freely, while others restrict their movement.
Isotonic solutions have the same concentration of solutes as the cells, resulting in no net movement of water. Hypotonic solutions have a lower concentration of solutes than the cells, causing water to move into the cells and potentially leading to cell swelling or bursting. Conversely, hypertonic solutions have a higher concentration of solutes than the cells, leading to water movement out of the cells and potential cell shrinkage or dehydration.
By understanding these key concepts, scientists and researchers can better interpret and explain various biological phenomena, such as the movement of molecules across cell membranes, the absorption of nutrients in the body, and the regulation of water balance in living organisms.
Diffusion and Osmosis: Similarities and Differences
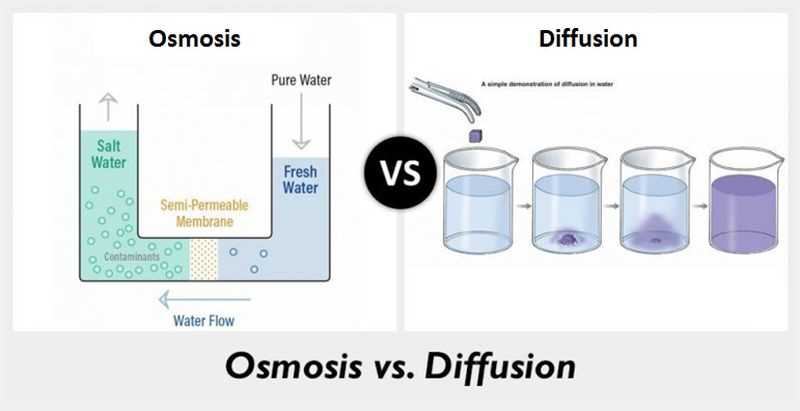
Diffusion and osmosis are both important processes that occur in biological systems. They are similar in that both involve the movement of molecules from an area of higher concentration to an area of lower concentration. This movement occurs passively, without the need for energy input from the cell or organism. However, there are also some key differences between diffusion and osmosis.
One key difference is that diffusion refers to the movement of any type of molecule, while osmosis specifically refers to the movement of water molecules. Diffusion can occur across a membrane or in a liquid, while osmosis typically occurs across a selectively permeable membrane, such as a cell membrane. In osmosis, water molecules move from an area of lower solute concentration to an area of higher solute concentration in an effort to equalize the concentration on both sides of the membrane.
Key Similarities:
- Both diffusion and osmosis involve the movement of molecules from an area of higher concentration to an area of lower concentration.
- Both processes occur passively, without the need for energy input.
Key Differences:
- Diffusion refers to the movement of any type of molecule, while osmosis specifically refers to the movement of water molecules.
- Diffusion can occur across a membrane or in a liquid, while osmosis typically occurs across a selectively permeable membrane.
- In osmosis, water molecules move from an area of lower solute concentration to an area of higher solute concentration.
In summary, diffusion and osmosis are similar in that they both involve the passive movement of molecules, but they differ in terms of the types of molecules involved and the types of membranes across which they occur.
Factors Affecting Diffusion and Osmosis
Diffusion and osmosis are two fundamental processes that play a crucial role in various biological and chemical systems. The rate and direction of diffusion and osmosis can be influenced by several factors.
Molecular Size: The size of the molecules or particles involved in diffusion or osmosis can significantly affect the rate at which they diffuse or osmose. Smaller molecules diffuse or osmose more quickly, as they can easily pass through the membrane or the spaces between other molecules.
Concentration Gradient: The concentration gradient, which is the difference in concentration between two regions, is another factor that affects the rate of diffusion or osmosis. The greater the concentration gradient, the faster the rate of diffusion or osmosis, as there is a higher driving force for molecules or particles to move from an area of high concentration to an area of low concentration.
Temperature: Temperature can also influence the rate of diffusion and osmosis. Higher temperatures generally increase the kinetic energy of molecules, making them move more vigorously. This increased motion can lead to faster diffusion or osmosis.
Surface Area: The surface area of the membrane or barrier through which diffusion or osmosis occurs is another important factor. A larger surface area allows for more molecules or particles to pass through at a given time, increasing the rate of diffusion or osmosis.
Distance: The distance that molecules or particles need to travel to reach their destination can also impact the rate of diffusion or osmosis. Generally, shorter distances result in faster diffusion or osmosis.
Overall, understanding these factors and how they affect diffusion and osmosis is important in various scientific fields, including biology, chemistry, and environmental sciences. By manipulating these factors, scientists can better control and predict the movement of molecules or particles in various systems.
Exploring Diffusion and Osmosis in Cells
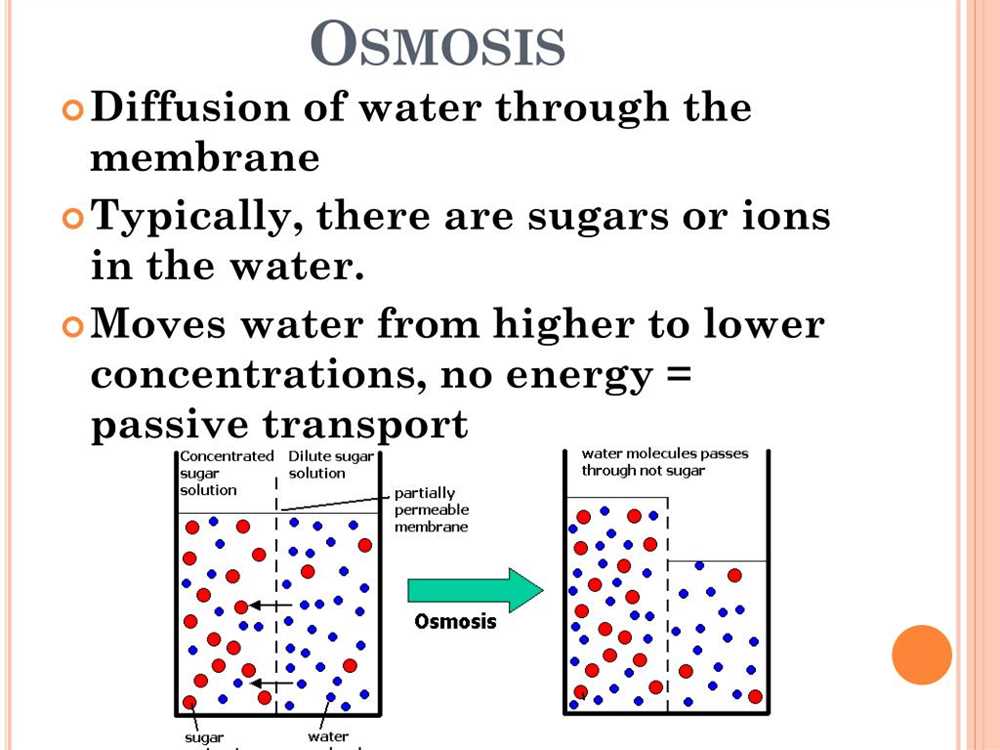
Diffusion and osmosis are fundamental processes that occur in cells to maintain homeostasis and enable proper functioning. Understanding these processes is crucial for comprehending how cells maintain their internal environment and exchange substances with their surroundings.
Diffusion is the spontaneous movement of particles from an area of higher concentration to an area of lower concentration. It occurs due to the random motion of particles and does not require any energy input. Diffusion plays a vital role in various cellular processes, such as the movement of gases, nutrients, and waste products across cell membranes.
Osmosis is a special type of diffusion that specifically refers to the movement of water molecules across a selectively permeable membrane. It occurs when there is a difference in solute concentration on either side of the membrane. Water molecules naturally move from an area of lower solute concentration to an area of higher solute concentration to maintain equilibrium.
One way to explore diffusion and osmosis in cells is through experiments. For example, a common laboratory activity involves placing a plant cell or an animal cell in a hypertonic (higher solute concentration) or hypotonic (lower solute concentration) solution and observing the changes in size and shape. In a hypertonic solution, water leaves the cell, causing it to shrink. In a hypotonic solution, water enters the cell, causing it to swell and potentially burst.
These experiments help us understand how cells react to different environments and how they regulate the movement of particles and water to maintain their internal balance. Diffusion and osmosis are essential processes that enable cells to survive and carry out their functions effectively.
Osmotic Pressure: Definition and Significance
Osmotic pressure is the pressure exerted by a solvent to prevent the net movement of a solvent across a semipermeable membrane. It is a fundamental concept in understanding the process of osmosis, which is the movement of solvent molecules from an area of lower solute concentration to an area of higher solute concentration through a semipermeable membrane. Osmotic pressure is the driving force behind osmosis and plays a crucial role in various biological and chemical processes.
The osmotic pressure of a solution depends on the concentration of solute particles in the solution. The greater the concentration of solute particles, the higher the osmotic pressure. This can be explained by the colligative properties of solutions, which are dependent on the number of solute particles rather than their identity or size. Osmotic pressure is directly related to the concentration of solute particles in a solution, making it a useful property to measure the concentration of solute in a solution.
Osmotic pressure has significant biological and industrial implications. In biological systems, osmotic pressure plays a vital role in maintaining cellular homeostasis. It regulates the movement of water and solutes across cell membranes, allowing cells to maintain proper hydration and nutrient balance. Osmotic pressure also affects the behavior of red blood cells, allowing them to maintain their shape and integrity in different osmotic environments.
In industrial applications, osmotic pressure is used in processes such as reverse osmosis, where it is exploited to separate solute and solvent molecules in water purification systems. Osmotic pressure also plays a significant role in processes such as dialysis and desalination. Understanding and manipulating osmotic pressure is crucial in these applications to achieve efficient separation and purification processes.
Solving Diffusion and Osmosis Problems: Step-by-Step Approach
Diffusion and osmosis are fundamental concepts in biology and understanding how they work is crucial for solving related problems. Whether you are studying for an exam or working on a research project, a step-by-step approach can help you solve these problems effectively.
1. Understanding the concepts: Before attempting to solve diffusion and osmosis problems, it is essential to have a clear understanding of the concepts involved. Diffusion is the movement of molecules from an area of high concentration to an area of low concentration, while osmosis is the diffusion of water across a selectively permeable membrane. Familiarize yourself with these definitions and their underlying principles.
2. Identify the problem: Read the problem carefully and identify the key information provided. Determine what exactly needs to be solved or calculated. Pay attention to values such as concentrations, volumes, and time. Identifying the problem correctly is crucial for selecting the appropriate formulas or equations to use.
3. Set up the problem: Once you understand the concepts and identify the problem, set up the problem by organizing the given information and converting units if necessary. This step helps you visualize the problem and determine which formula or equation to use to solve it. This may involve calculating concentrations, volumes, or masses.
4. Apply the appropriate equations: Use the appropriate formulas or equations to solve the problem. For example, Fick’s first law of diffusion can be used to calculate the rate of diffusion, while the van’t Hoff Equation can be used to calculate osmotic pressure. Make sure to use the correct units and variables when plugging in the values.
5. Solve the equations: Once you have applied the appropriate equations, solve them step by step. This may involve rearranging equations, canceling out units, or simplifying expressions. Pay attention to significant figures and round the final answer to the appropriate decimal places.
6. Check your answer: After solving the equations, double-check your answer to ensure its accuracy. Compare it with the given information and see if it makes sense in the context of the problem. If necessary, re-evaluate your calculations or review the concepts to ensure you have arrived at the correct answer.
By following this step-by-step approach, you can effectively solve diffusion and osmosis problems in biology. Practice with different problems and seek clarification if you encounter any difficulties. With time and practice, you will become comfortable solving these types of problems and gain a deeper understanding of diffusion and osmosis.