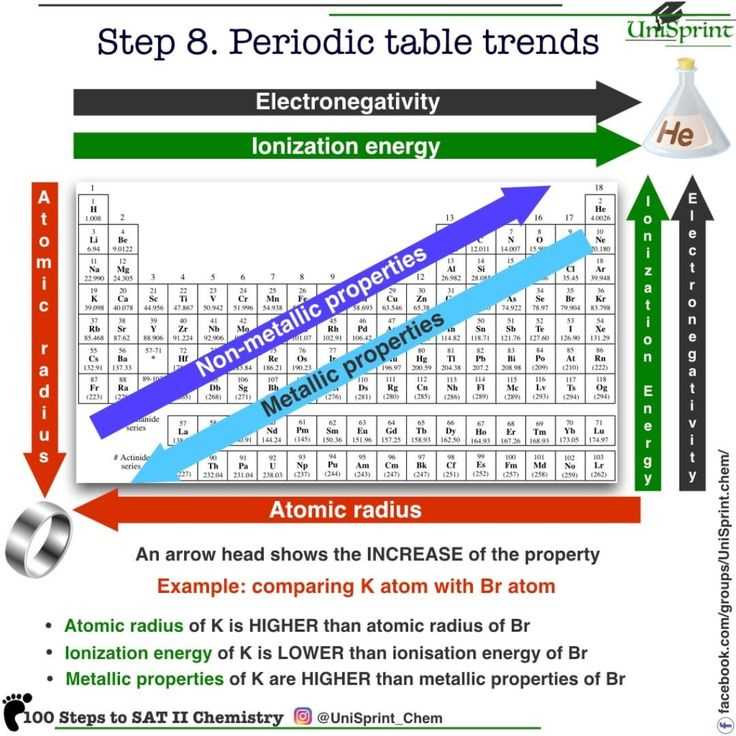
Understanding the periodic trends in the periodic table is essential for predicting the properties of elements. These trends allow us to make predictions about an element’s atomic size, ionization energy, electronegativity, and reactivity. In this article, we will review the answers to some common questions about periodic trends.
Firstly, the atomic size trend refers to how the size of an atom changes as you move across or down the periodic table. It is generally observed that atomic size increases as you move down a group and decreases as you move across a period. This is due to the increasing number of energy levels as you move down a group, leading to larger atoms. Conversely, as you move across a period, the atomic size decreases because the increasing number of protons draws the electron cloud closer to the nucleus, making the atom smaller.
Secondly, ionization energy is the energy required to remove an electron from an atom. The trend for ionization energy is generally observed to increase as you move across a period and decrease as you move down a group. This is because the increasing number of protons in the nucleus leads to a greater attraction for the electrons, making it harder to remove them. Conversely, as you move down a group, the atomic size increases, making it easier to remove an electron.
Lastly, electronegativity refers to an atom’s ability to attract electrons towards itself in a chemical bond. The trend for electronegativity is observed to increase as you move across a period and decrease as you move down a group. This is due to the same reasons as ionization energy – the increasing number of protons leads to a greater attraction for electrons, making an atom more electronegative. Conversely, as you move down a group, the atomic size increases, making it less likely for an atom to attract electrons.
Understanding and recognizing these periodic trends can help predict and explain the chemical behavior and properties of elements. By analyzing the periodic table, scientists are able to make educated guesses about an element’s behavior, even if they have never observed or studied it before. Periodic trends are a fundamental concept in chemistry and are an essential tool for understanding the world of elements and their properties.
What are periodic trends?

Periodic trends refer to the patterns and variations in properties of elements that occur as you move across a period or down a group on the periodic table. These trends are important in understanding the behavior and characteristics of elements, as they provide valuable information about their atomic structure and chemical reactivity.
One of the most well-known periodic trends is atomic radius, which describes the size of an atom. As you move down a group, the atomic radius generally increases due to the addition of new energy levels. For example, in Group 1 (alkali metals), the atomic radius increases as you go from lithium to cesium. On the other hand, as you move across a period from left to right, the atomic radius generally decreases due to the increasing nuclear charge pulling the electrons closer to the nucleus. This can be observed in Period 3, where the atomic radius decreases from sodium to chlorine.
Another important periodic trend is electronegativity, which measures an atom’s ability to attract and bond with electrons. Electronegativity generally increases across a period and decreases down a group. Oxygen, with an electronegativity of 3.44, is one of the most electronegative elements, whereas elements like cesium, with an electronegativity of 0.79, have a lower electronegativity value.
Periodic trends also include ionization energy, which is the energy required to remove an electron from an atom, and electron affinity, which is the energy change when an atom gains an electron. These trends provide insight into an element’s stability, reactivity, and bonding behavior. By understanding periodic trends, scientists can make predictions and explanations about the properties and behaviors of different elements and their compounds.
Periodic Trends in Atomic Radius
The atomic radius is a measure of the size of an atom. It refers to the distance between the nucleus of an atom and its outermost electron. Understanding the periodic trends in atomic radius can provide valuable insights into the properties and behavior of elements.
One key trend in atomic radius is that it generally decreases from left to right across a period of the periodic table. This is known as the period trend. This trend occurs because as you move from left to right across a period, the number of protons in the nucleus increases, resulting in a stronger attraction between the nucleus and the outermost electrons. The increased attraction pulls the electrons closer to the nucleus, making the atomic radius smaller.
An opposite trend can be observed when moving down a group in the periodic table. This is known as the group trend. The atomic radius generally increases as you move down a group. This is because as you move down a group, the number of electron shells increases. This means there are more energy levels for the electrons to occupy, leading to an overall larger atomic radius.
It is important to note that exceptions to these trends can occur due to specific electron configurations or other factors. For example, the atomic radius of transition metals can vary within a group due to the addition of inner d electron orbitals. Additionally, factors such as ionic radii and the presence of electron clouds can also influence atomic radius.
In conclusion, understanding the periodic trends in atomic radius provides important insights into the size and behavior of atoms. The period trend shows a decrease in atomic radius from left to right across a period, while the group trend shows an increase in atomic radius as you move down a group. While there may be exceptions to these trends, they serve as a valuable tool for understanding the properties of elements.
Periodic trends in ionization energy
The ionization energy is defined as the energy required to remove one electron from an atom or ion. This property of an atom is influenced by various factors, including atomic size, electron shielding, and nuclear charge. By examining the periodic table, we can observe certain trends in ionization energy.
Firstly, as we move across a period from left to right, the ionization energy generally increases. This can be attributed to the increase in nuclear charge, which attracts the electrons more strongly, making it more difficult to remove an electron. Additionally, as we move from left to right across a period, the atomic size decreases, leading to a stronger pull on the outermost electrons by the nucleus.
Example: The ionization energy of sodium (Na) is lower than that of magnesium (Mg) in the same period. This is because sodium has a larger atomic size and a lower nuclear charge compared to magnesium, making it easier to remove an electron from sodium.
Secondly, as we move down a group, the ionization energy generally decreases. This is due to the increase in atomic size and the shielding effect of inner core electrons. The larger distance between the nucleus and outermost electrons in larger atoms results in a weaker pull on the electrons, making it easier to remove an electron.
Example: The ionization energy of lithium (Li) is lower than that of beryllium (Be) in the same group. This is because lithium has a larger atomic size and more inner core electrons, leading to a weaker attraction between the nucleus and the outermost electron.
In summary, the ionization energy generally increases across a period and decreases down a group. This trend can be explained by the interplay of atomic size, electron shielding, and nuclear charge in determining the ease of removing an electron from an atom.
Periodic trends in electron affinity

Electron affinity is a measure of how readily an atom gains an extra electron to form a negative ion. It is an important property that helps us understand the chemical behavior of elements. Electron affinity tends to increase across a period from left to right on the periodic table. This means that atoms on the right side of the periodic table have higher electron affinities, as they are more likely to gain an electron due to their relatively small size and high effective nuclear charge.
A key trend in electron affinity is the jump in values between Group 15 (Nitrogen) and Group 16 (Oxygen) elements. Nitrogen has a slightly positive electron affinity, meaning it is less likely to gain an electron. Oxygen, on the other hand, has a significantly higher electron affinity, making it more likely to gain an electron. This jump occurs because the addition of an electron to Group 16 elements leads to the formation of a stable, filled p orbital, resulting in a more favorable electron configuration.
Another trend in electron affinity can be observed when moving down a group. As we move down the periodic table, the electron affinity generally decreases. This is due to an increase in atomic size and screening effect, which reduces the attractive force between the nucleus and the incoming electron. As a result, atoms in lower periods are less likely to gain an electron, leading to lower electron affinity values.
Overall, the study of electron affinity provides valuable insights into the reactivity and chemical behavior of elements. By understanding these trends, we can predict how elements will react and form compounds, allowing us to further explore the amazing world of chemistry.
Periodic trends in electronegativity

Electronegativity is a measure of an atom’s ability to attract electrons in a chemical bond. It is an important concept in understanding the properties of elements and their behavior in chemical reactions. The electronegativity of an element can vary across the periodic table, showing distinct trends and patterns.
One of the periodic trends in electronegativity is that it generally increases from left to right across a period. This means that elements on the right side of the periodic table tend to have higher electronegativities compared to elements on the left side. This trend can be attributed to the increase in effective nuclear charge as the number of protons in the nucleus increases. The stronger the positive charge of the nucleus, the more it pulls the electrons towards itself, resulting in higher electronegativity.
Another periodic trend in electronegativity is that it generally decreases down a group. Elements in the same group have similar outer electron configurations, which means they have similar electron-electron repulsions. This similarity in electron-electron repulsions leads to a decrease in electronegativity as you move down a group. Additionally, the increase in atomic size down a group also plays a role in decreasing electronegativity. The larger distance between the nucleus and the outer electrons weakens the attractive force and reduces the electronegativity.
Understanding the periodic trends in electronegativity is essential in predicting the nature of chemical bonds and the behavior of elements in various compounds. These trends provide valuable insights into the characteristics of elements and help in explaining their reactivity and bonding properties.
Periodic trends in metallic character
The metallic character of elements refers to their ability to conduct electricity and heat. Generally, metals are good conductors of both electricity and heat, while nonmetals are poor conductors. The metallic character of an element is influenced by several periodic trends, including atomic radius, ionization energy, and electronegativity.
One of the main factors influencing the metallic character is atomic radius. As you move down a group on the periodic table, the atomic radius increases due to the addition of more electron shells. This leads to increased metallic character because the outer electrons are further from the nucleus and therefore more easily released to conduct electricity.
Another factor affecting metallic character is ionization energy. Ionization energy refers to the energy required to remove an electron from an atom. Generally, metals have low ionization energies because their outer electrons are loosely bound. This makes it easier for the metal atoms to lose electrons and form positive ions, which are essential for conducting electricity.
Electronegativity, the tendency of an atom to attract a shared pair of electrons in a covalent bond, also plays a role in determining the metallic character. Metals generally have low electronegativities because they have a tendency to lose electrons rather than gain them. This property allows them to form positive ions and exhibit metallic behavior.
In summary, the metallic character of an element is influenced by atomic radius, ionization energy, and electronegativity. Elements with larger atomic radii, lower ionization energies, and lower electronegativities tend to have greater metallic character. Understanding these periodic trends can help predict the properties and behavior of elements in terms of their metallic character.
Factors influencing periodic trends
The periodic table of elements displays a variety of trends, or patterns, that can be observed among the elements. These trends are influenced by several factors.
One factor that affects periodic trends is the atomic size or radius. The atomic radius is the distance from the nucleus to the outermost shell of electrons. As you move from left to right across a period on the periodic table, the atomic radius generally decreases. This is because the nuclear charge increases, pulling the electrons closer to the nucleus. On the other hand, as you move down a group, the atomic radius generally increases. This is because the number of electron shells increases, resulting in increased distance between the outermost electrons and the nucleus.
- Electron shielding: Another factor that influences periodic trends is electron shielding. Electron shielding refers to the repulsion between electrons in different energy levels. When electrons occupy inner energy levels, they shield the outer electrons from the full effect of the nuclear charge. This leads to an increase in atomic radius as you move down a group.
- Ionization energy: Ionization energy is the energy required to remove an electron from an atom or ion in the gaseous state. Ionization energy generally increases as you move from left to right across a period. This is because the atomic radius decreases, making it more difficult to remove an electron. On the other hand, ionization energy generally decreases as you move down a group, as the outermost electrons are farther from the nucleus and are therefore easier to remove.
- Electronegativity: Electronegativity is a measure of an atom’s ability to attract shared electrons in a chemical bond. Electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. This is due to the increasing nuclear charge and decreasing atomic size.
In summary, periodic trends are influenced by factors such as atomic size, electron shielding, ionization energy, and electronegativity. These factors contribute to the patterns observed in the periodic table and help us understand the behavior and properties of the elements.