
A phase diagram is a graphical representation of the different phases of a substance under various conditions of temperature and pressure. It is a valuable tool in understanding the behavior of materials and predicting their phase transitions. Phase diagrams can help scientists and engineers design and optimize processes and materials for specific applications.
In Phase Diagram Worksheet 2, students are provided with a diagram and asked to answer questions related to the phases and phase transitions of a substance. By analyzing the diagram and applying their knowledge of phase diagrams, students can determine the melting point, boiling point, critical point, and other key properties of the substance.
The answers to the worksheet provide students with a better understanding of the relationships between temperature, pressure, and the different phases of a substance. By working through the worksheet and comparing their answers with the provided solutions, students can reinforce their understanding of phase diagrams and enhance their problem-solving skills in this area.
Overall, Phase Diagram Worksheet 2 and its answers serve as a valuable educational resource for students studying materials science, chemistry, or any other field that involves the characterization and manipulation of materials. It provides a hands-on approach to learning about phase diagrams and enables students to apply their knowledge to real-world scenarios. Through this worksheet, students can develop a deeper understanding of phase transitions and their relevance in various scientific and industrial contexts.
Phase Diagram Worksheet 2 Answers: Explained and Simplified
In the field of chemistry, understanding phase diagrams is essential for predicting and explaining the behavior of substances under different conditions. Phase diagrams provide us with a graphical representation of the equilibrium phases and their transitions as temperature and pressure change. The Phase Diagram Worksheet 2 offers a set of questions focused on interpreting and analyzing phase diagrams, and learning the various terminologies associated with them.
One of the key concepts covered in the worksheet is the identification of the different phases present at specific temperature and pressure values. Answering the questions requires the application of knowledge about the characteristics of each phase and their corresponding boundaries on the phase diagram. By comprehending the information presented on the diagram and analyzing the given values, students can determine the phases involved in a particular scenario.
To effectively answer the questions, it is crucial to understand the definitions of terms like vapor pressure, critical point, triple point, and phase boundaries. The worksheet provides the opportunity to practice identifying these key points on the phase diagram and relate them to the corresponding physical properties of substances. Additionally, the worksheet challenges students to explain the behavior of substances in terms of their phase transitions, including melting, boiling, and sublimation.
The Phase Diagram Worksheet 2 answers play an important role in the learning process by providing explanations and solutions to the given questions. Students are able to check their understanding and verify their own answers by referring to the provided answers. This feedback allows them to identify any misconceptions they may have and seek further clarification if necessary. Through this iterative process of exploration and correction, students can solidify their understanding of phase diagrams and improve their problem-solving skills in the field of chemistry.
Understanding the Basics of Phase Diagrams
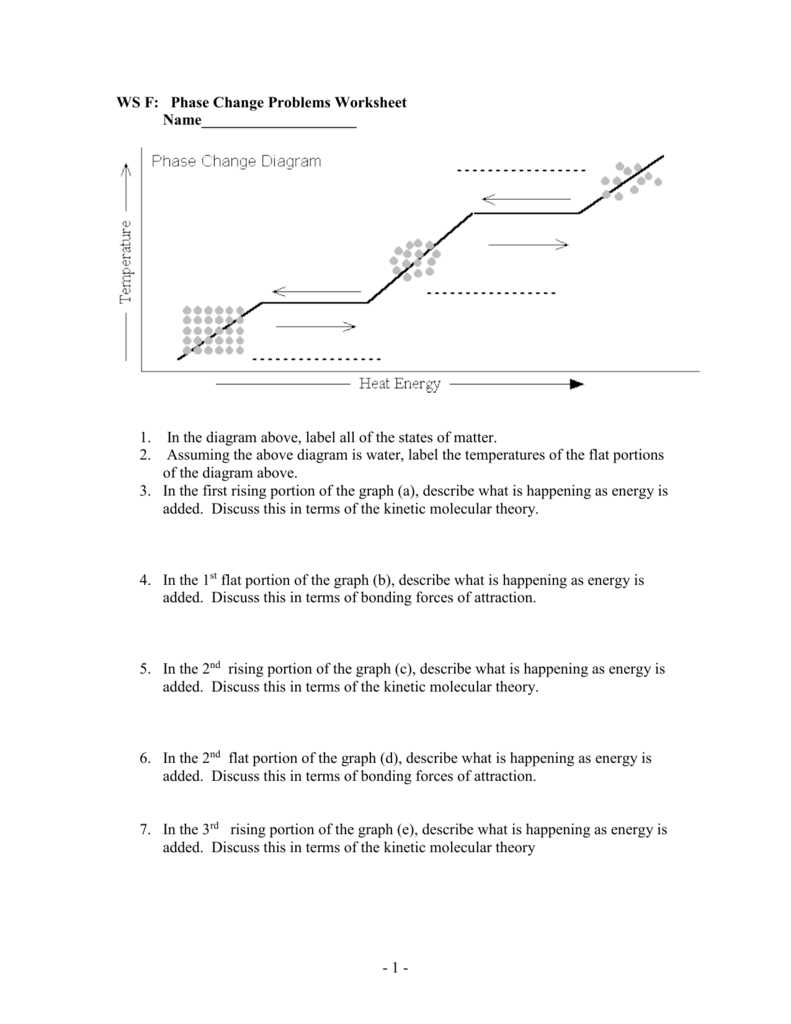
A phase diagram is a graphical representation of the different phases of a substance, such as solid, liquid, and gas, as a function of temperature and pressure. It is a valuable tool used by scientists and engineers to understand and predict the behavior of materials under different conditions.
Phase boundaries: At any given temperature and pressure, a substance can exist in different phases. The phase diagram shows the boundaries that separate these phases from each other. The lines on the diagram represent the points where these phase transitions occur.
For example, let’s consider the phase diagram of water. At low temperatures and pressures, water exists in the solid phase, represented by the line separating the solid (ice) and liquid phases. As temperature and pressure increase, water transitions to the liquid phase, represented by the line separating the liquid and gas phases. At higher temperatures and pressures, water exists in the gaseous phase.
Triple point: The phase diagram also includes a point called the triple point, which is the unique combination of temperature and pressure at which all three phases of a substance coexist in equilibrium. In the case of water, the triple point occurs at 0.01 degrees Celsius and 611.657 pascals (Pa).
Phase diagrams for mixtures: Phase diagrams are not only useful for pure substances but also for mixtures. In the case of mixtures, the phase diagram shows how the proportions of different components affect the phase behavior. For example, the phase diagram of a binary mixture (two-component system) would show how the mixture changes from solid to liquid to gas as the composition of the components varies.
Understanding phase diagrams is essential for various fields of science and engineering, such as material science, thermodynamics, and chemical engineering. These diagrams provide valuable insights into the properties and behavior of substances, helping researchers design and optimize processes that involve phase transitions.
Importance of Phase Diagram Worksheet 2
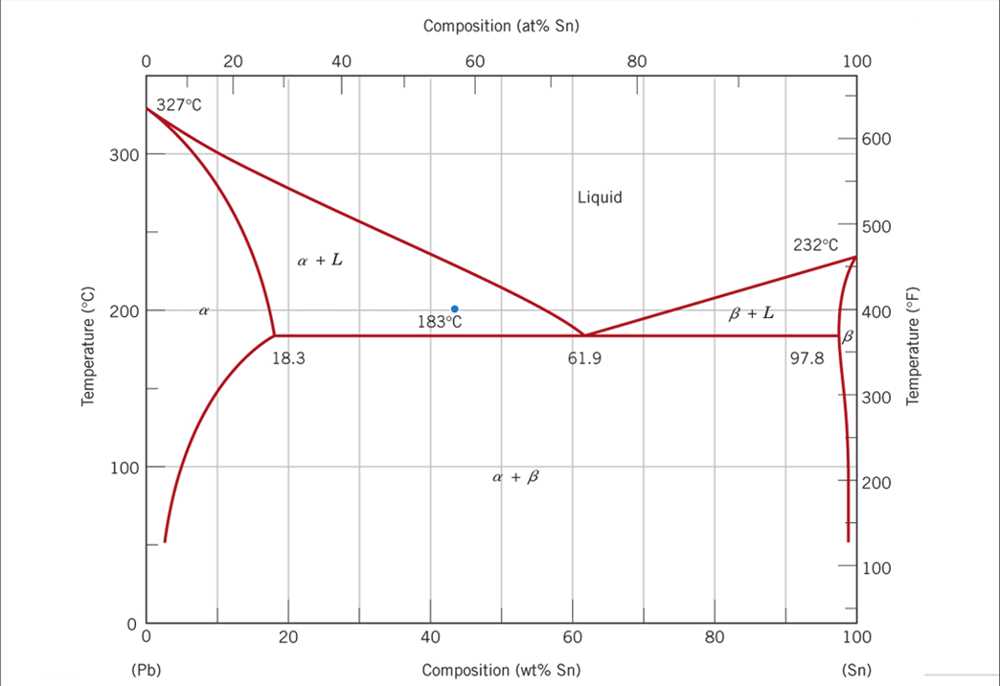
Phase diagrams are essential tools in the field of physical chemistry, as they provide a visual representation of the different phases of a substance at various temperatures and pressures. They help scientists and engineers understand and predict the behavior of materials and optimize processes in industries such as pharmaceuticals, materials science, and chemical engineering.
Phase Diagram Worksheet 2 is an important tool for students learning about phase diagrams. It offers a practical and hands-on approach to understanding the concepts and principles behind phase diagrams. By completing the worksheet, students can practice interpreting phase diagrams, identifying phase boundaries, and predicting changes in phase behavior based on temperature and pressure variations.
The worksheet contains a variety of exercises and questions that challenge students to apply their knowledge and problem-solving skills. It encourages them to think critically, analyze data, and make connections between theoretical concepts and real-world applications. Through this process, students develop a deeper understanding of phase diagrams and their significance in the field of physical chemistry.
Furthermore, Phase Diagram Worksheet 2 allows students to practice drawing and constructing phase diagrams themselves. This hands-on experience helps reinforce their understanding of the relationships between temperature, pressure, and phase transitions. It also helps them develop important skills in data visualization and graphical representation, which are valuable in many scientific and engineering disciplines.
In conclusion, Phase Diagram Worksheet 2 is a valuable tool for students studying physical chemistry. It helps them develop a solid understanding of phase diagrams and their applications, while also honing their problem-solving and data interpretation skills. By engaging with the worksheet, students can gain a deeper appreciation for the importance of phase diagrams in various scientific and engineering fields.
Key Concepts Covered in Phase Diagram Worksheet 2
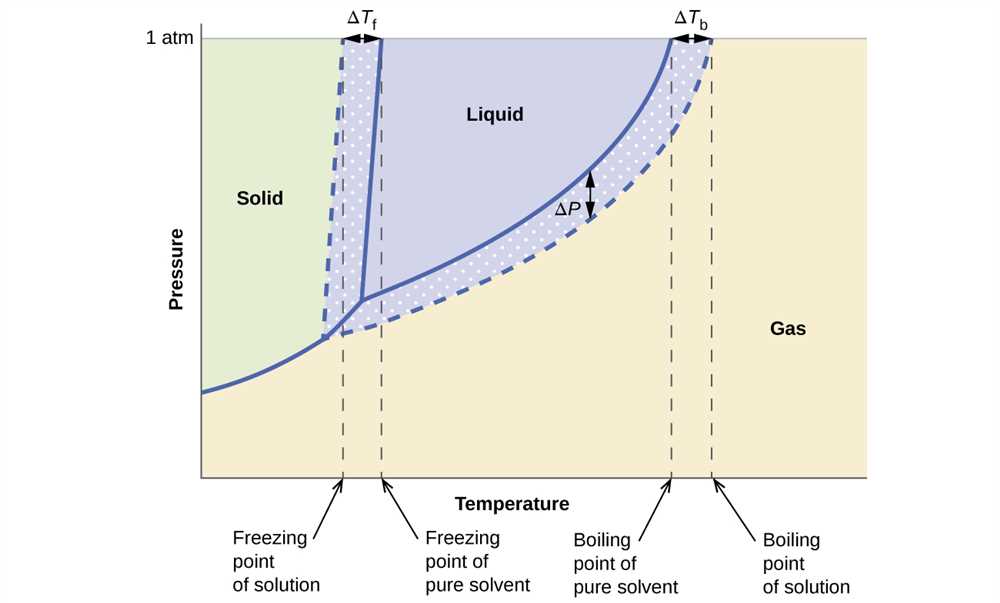
Phase Diagram Worksheet 2 covers several important concepts related to phase diagrams and the behavior of substances at different temperatures and pressures. By completing this worksheet, students can deepen their understanding of these key concepts and gain a clearer picture of how various substances behave under different conditions.
Some of the key concepts covered in Phase Diagram Worksheet 2 include:
- Phase Diagrams: Students will learn how to interpret phase diagrams, which visually represent the different phases (solid, liquid, gas) that a substance can exist in at different temperatures and pressures. They will become familiar with the different regions on a phase diagram and the boundaries between the phases.
- Phase Transitions: The worksheet explores different types of phase transitions, such as melting, boiling, and sublimation. Students will learn how these transitions occur at specific temperatures and pressures, and how they can be influenced by changing these conditions.
- Critical Points: The worksheet introduces the concept of the critical point, which represents the temperature and pressure at which a substance undergoes a phase transition from a liquid to a gas without distinct boundaries between the phases. Students will learn about the critical point on a phase diagram and its significance.
- Triple Points: The concept of triple points is covered in the worksheet, which refers to the temperature and pressure at which all three phases of a substance (solid, liquid, gas) coexist in equilibrium. Students will understand the significance of triple points and how they are represented on a phase diagram.
- Phase Diagram Calculations: The worksheet also includes practical calculations based on phase diagrams, such as determining the temperature and pressure at which a substance transitions from one phase to another, or calculating the composition of a mixture of two substances at a given temperature and pressure.
By exploring these key concepts through Phase Diagram Worksheet 2, students can develop a solid foundation in understanding phase diagrams and the behavior of substances under different conditions. This knowledge is vital in various scientific fields, such as chemistry, materials science, and engineering.
Analyzing Phase Diagram Worksheet 2: Sample Questions
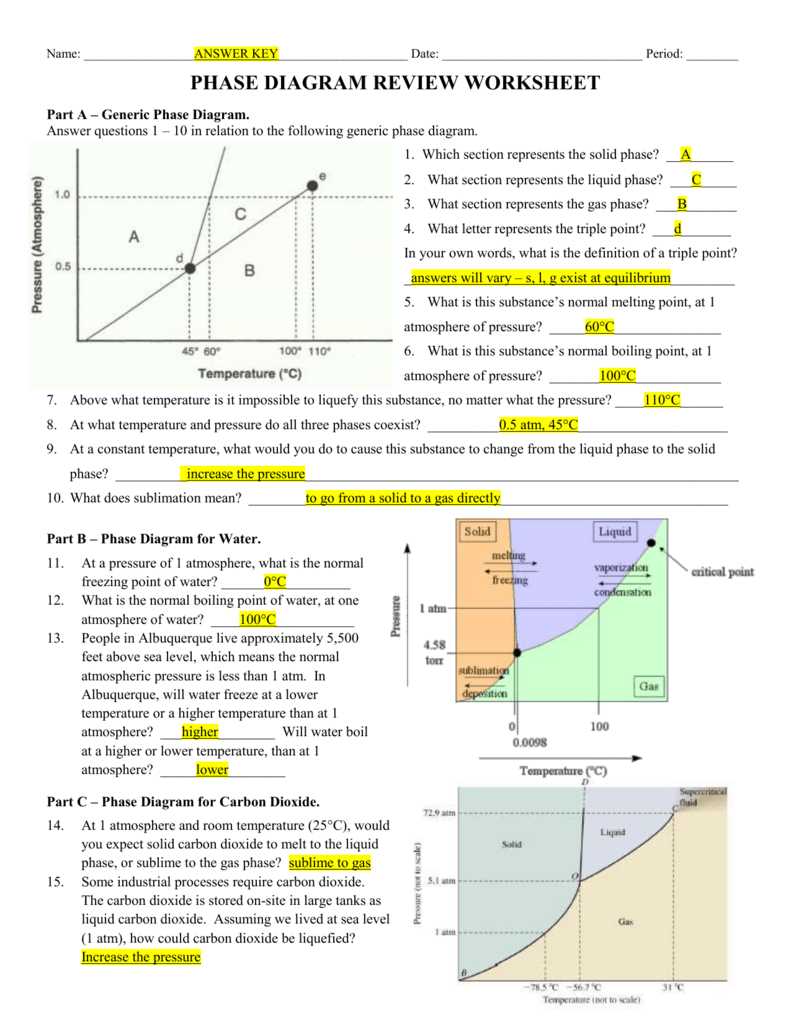
In Phase Diagram Worksheet 2, students are presented with a series of questions designed to test their understanding of phase diagrams and the relationship between temperature and pressure in determining the state of a substance. These questions cover a range of topics, including interpreting phase diagrams, identifying phase transitions, and determining the conditions under which different phases coexist.
One example question asks students to analyze a phase diagram and identify the temperature and pressure at which a substance is in a solid, liquid, or gas state. This question requires students to use their knowledge of phase diagrams and the properties of different phases to determine the correct answer. It also challenges students to think critically and apply their understanding of the relationship between temperature and pressure in determining the state of a substance.
Another question asks students to interpret a phase diagram and determine the conditions under which a substance can exist in equilibrium between two phases. This question requires students to analyze the diagram and identify the points where the lines representing different phases intersect. By doing so, students can determine the temperature and pressure at which the substance can coexist in both phases simultaneously. This question tests students’ ability to interpret phase diagrams and understand the concept of phase equilibrium.
Overall, Phase Diagram Worksheet 2 provides students with an opportunity to apply their knowledge of phase diagrams and the relationship between temperature and pressure in a variety of contexts. By analyzing and answering the sample questions, students can reinforce their understanding of these fundamental concepts and develop their skills in interpreting phase diagrams and identifying phase transitions. The worksheet serves as a valuable tool for both assessing students’ comprehension and facilitating their learning in this important area of physical chemistry.
Step-by-Step Solutions for Phase Diagram Worksheet 2
If you are looking for step-by-step solutions for Phase Diagram Worksheet 2, you have come to the right place. In this guide, we will go through each problem and provide detailed explanations for each step. By following along, you will gain a better understanding of phase diagrams and be able to confidently solve similar problems in the future.
Problem 1: The problem asks you to interpret a phase diagram and determine the state of a substance at certain temperatures and pressures. To solve this, start by locating the given temperature and pressure values on the phase diagram. Then, identify the corresponding phase for each point. The key on the diagram will help you determine whether the substance is in the solid, liquid, or gas phase.
Problem 2: This problem requires you to calculate the boiling point and melting point of a substance using the phase diagram. Begin by locating the triple point on the diagram, which represents the temperature and pressure at which all three phases of the substance coexist. From there, use the phase boundaries to determine the melting and boiling points. The melting point is where the solid and liquid phases intersect, while the boiling point is where the liquid and gas phases intersect.
To help you further, we have compiled a list of key terms and concepts commonly encountered in phase diagram problems:
- Phase boundary: the line on the diagram separating two phases
- Triple point: the temperature and pressure at which all three phases of a substance coexist
- Critical point: the temperature and pressure above which the substance cannot exist in a liquid state
- Sublimation: the process in which a solid directly turns into a gas without going through the liquid phase
- Condensation: the process in which a gas turns into a liquid
By understanding these terms and concepts, you will be better equipped to solve phase diagram problems. Remember to carefully study the given information, locate the points on the diagram, and interpret the phase changes correctly. With practice, you will become more proficient in analyzing and solving phase diagram problems.