
Understanding potential energy diagrams is crucial in chemistry as they help us visualize the energy changes that occur during chemical reactions. These diagrams show the energy levels of reactants and products, as well as the energy barriers that need to be overcome for a reaction to occur.
In this potential energy diagram worksheet, you will find answers that will guide you through the process of analyzing and interpreting potential energy diagrams. By understanding these diagrams, you will be able to predict whether a reaction is exothermic or endothermic, identify transition states and activation energies, and determine the overall energy change of a reaction.
The worksheet answers will provide step-by-step explanations on how to calculate the energy change between reactants and products, and how to identify the intermediates and transition states in a reaction. It will also help you understand the relationship between bond energies and potential energy, and how changes in molecular structure affect the energy of a molecule.
By working through this potential energy diagram worksheet and understanding the answers, you will gain a deeper insight into the role of energy in chemical reactions. You will be able to apply this knowledge to predict the feasibility of reactions, understand reaction mechanisms, and make informed decisions in various chemical processes.
Potential Energy Diagram Worksheet Answers
In the study of chemistry, potential energy diagrams are often used to represent the changes in energy that occur during a chemical reaction. These diagrams show the potential energy of a system over the course of the reaction, with the reactants on the left and the products on the right.
One common question that arises when studying potential energy diagrams is how to interpret the different features of the diagram. For example, the activation energy can be identified as the energy barrier that must be overcome for the reaction to proceed. This energy barrier is represented by the highest point on the diagram, known as the transition state or activated complex.
Another important feature of a potential energy diagram is the difference in energy between the reactants and the products, known as the overall energy change or the heat of reaction. This energy change can be determined by subtracting the energy of the reactants from the energy of the products, and it indicates whether the reaction is exothermic (releases heat) or endothermic (absorbs heat).
Understanding potential energy diagrams is crucial for predicting the feasibility and energetics of chemical reactions. By analyzing the diagram and calculating the energy changes involved, scientists can determine the stability of reactants and products, as well as the pathway and rate of the reaction. This information is valuable for designing and optimizing chemical processes and reactions in various industries, including pharmaceuticals, materials science, and environmental engineering.
Understanding Potential Energy Diagrams

In chemistry and physics, potential energy diagrams are graphical representations of the energy changes that occur during a chemical reaction or a physical process. These diagrams help us understand the relative energy levels of the reactants and products, as well as the energy barriers that need to be overcome for the reaction to occur.
One key element of a potential energy diagram is the vertical axis, which represents the potential energy of the system. The reactants are usually placed on the left side, while the products are placed on the right side. The curves or lines connecting the reactants and products represent the energy changes that occur during the reaction. These can be either continuous curves or step-like curves, depending on the nature of the reaction.
Another important feature of potential energy diagrams is the presence of energy barriers or activation energies. These barriers represent the minimum amount of energy that the reactants need to overcome in order for the reaction to proceed. If the reactants do not have enough energy to surpass these barriers, the reaction will not occur. The height of these barriers can provide information about the speed of the reaction; higher barriers indicate slower reactions.
In addition to reactants and products, potential energy diagrams may also include intermediate states or transition states. These states represent temporary arrangements of atoms or molecules that occur during the reaction but do not exist as stable products. The energy levels of these intermediate states can be higher or lower than the energy levels of the reactants and products.
In conclusion, potential energy diagrams provide a visual representation of the energy changes that occur during chemical reactions or physical processes. By analyzing these diagrams, scientists can understand the relative energy levels, energy barriers, and reaction pathways. This understanding is crucial for designing and optimizing various chemical processes and reactions.
Exploring the Concept of Potential Energy

Potential energy is a fundamental concept in physics that refers to the energy possessed by an object due to its position or condition. It is a form of energy that can be stored and converted into other forms, such as kinetic energy, which is the energy of an object in motion. Understanding potential energy is crucial in many areas of science and engineering, as it helps explain various phenomena and allows for the prediction and analysis of systems.
There are different types of potential energy, including gravitational potential energy, elastic potential energy, and chemical potential energy. Gravitational potential energy is associated with an object’s height above the ground and its mass. As an object is lifted higher, its potential energy increases. Elastic potential energy is related to the deformation of an object, such as a stretched spring or compressed rubber band. Chemical potential energy is stored in the atoms and molecules that make up substances and can be released during chemical reactions.
Gravitational potential energy can be calculated using the formula: PE = mgh, where m is the mass of the object, g is the acceleration due to gravity, and h is the height of the object above a reference point. Elastic potential energy can be calculated using the formula: PE = (1/2)kx^2, where k is the spring constant and x is the displacement of the spring from its equilibrium position.
Potential energy diagrams are graphical representations that show the changes in potential energy as a system undergoes a transformation or process. They are useful tools for visualizing and analyzing the energy changes that occur in chemical reactions, phase transitions, and other physical and chemical processes. These diagrams typically plot potential energy on the y-axis and a reaction coordinate or progress of the process on the x-axis.
Overall, potential energy is a concept that plays a crucial role in understanding the behavior of objects and systems. It enables scientists and engineers to analyze and predict the outcomes of various processes and transformations. By studying potential energy, we can gain insights into the fundamental principles of energy conservation and the interplay between different forms of energy.
Analyzing the Shapes of Potential Energy Diagrams
Potential energy diagrams are graphical representations that depict the changes in potential energy as a reaction progresses. These diagrams provide valuable insight into the energy changes that occur during a chemical reaction and can help us understand the overall behavior of the system.
One key aspect of analyzing potential energy diagrams is examining the shapes of the curves. The shape of the curve indicates the energy changes that are taking place within the system. For example, a curve that starts at a high energy level and gradually decreases represents an exothermic reaction, where energy is released. On the other hand, a curve that starts at a low energy level and gradually increases represents an endothermic reaction, where energy is absorbed.
Another important feature to consider is the presence of energy barriers or activation energy. These barriers are represented by energy peaks on the diagram. They indicate the minimum energy required for the reaction to occur and the rate at which the reaction proceeds. The height of the energy barrier determines the reaction rate – higher barriers generally correspond to slower reactions.
Additionally, the transition state or activated complex is also depicted on the potential energy diagram. This is the point at which the reactants are in an unstable state and are on the verge of forming products. The transition state is represented by the highest point on the energy curve and is a crucial point in understanding the reaction mechanism.
Summary:
- Potential energy diagrams show the changes in potential energy during a reaction.
- The shape of the curve indicates the energy changes and whether the reaction is exothermic or endothermic.
- Energy barriers represent the minimum energy required for the reaction to occur.
- The transition state is the highest point on the energy curve and represents the reactants in an unstable state.
Identifying Key Points on Potential Energy Diagrams
Potential energy diagrams are graphical representations that show the changes in potential energy as reactants turn into products during a chemical reaction. These diagrams are essential for understanding the energy changes and the activation energy of a reaction. By identifying key points on a potential energy diagram, we can gather important information about the reaction.
1. Reactants: The reactants are the starting materials of a chemical reaction. On a potential energy diagram, they are represented by the initial energy level. It is the lowest point on the diagram and corresponds to the potential energy of the reactants before the reaction takes place.
2. Products: The products are the end result of a chemical reaction. They are represented by the highest energy level on the potential energy diagram. This point shows the potential energy of the products after the reaction is complete.
3. Activation energy: Activation energy is the energy barrier that must be overcome for a chemical reaction to occur. It is represented by the peak on the potential energy diagram. The higher the peak, the higher the activation energy required for the reaction to proceed.
4. Transition state: The transition state is the intermediate structure formed during the chemical reaction. It is represented by the highest point on the potential energy diagram, also known as the activated complex. The transition state has higher potential energy than both the reactants and the products.
5. Exothermic and endothermic reactions: An exothermic reaction releases energy, resulting in a potential energy diagram with the products at a lower energy level than the reactants. On the other hand, an endothermic reaction absorbs energy, causing the products to have a higher energy level than the reactants on the diagram.
By understanding and identifying these key points on potential energy diagrams, scientists can gather crucial information about the energy changes and the progress of a chemical reaction. This knowledge can help in designing and optimizing reactions for various applications in fields such as pharmaceuticals, materials science, and energy production.
Determining Activation Energy and Reaction Rates
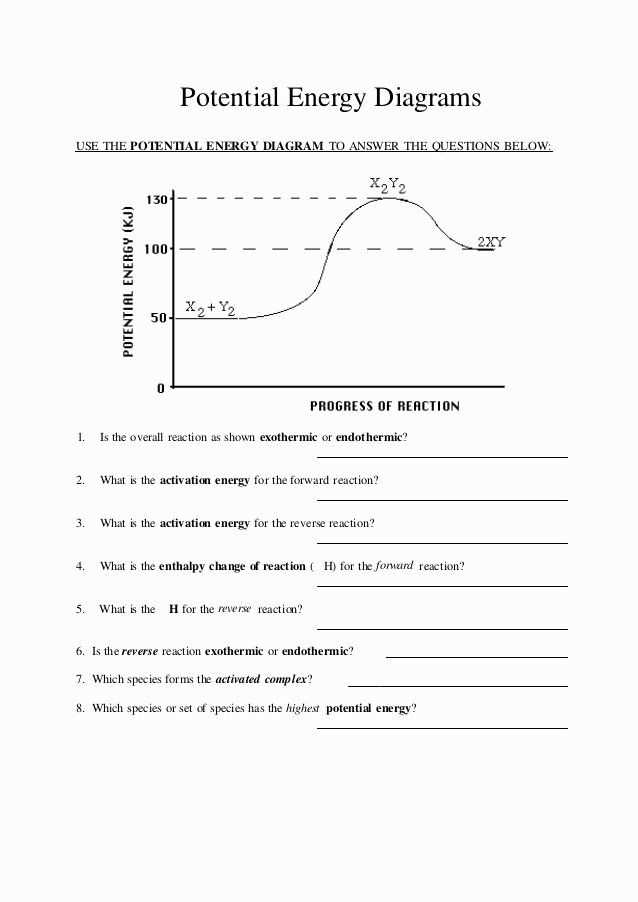
In chemistry, activation energy refers to the minimum amount of energy required for a chemical reaction to occur. It represents the energy barrier that must be overcome in order for reactant molecules to be transformed into product molecules. Understanding the activation energy of a reaction is crucial for predicting the speed at which the reaction will proceed.
To determine the activation energy of a reaction, scientists often use the Arrhenius equation. This equation relates the rate constant of a reaction with the temperature and activation energy. By measuring the rate constant at different temperatures and plotting a graph of ln(rate constant) versus 1/T, the activation energy can be calculated using the slope of the line.
In order to accurately determine the activation energy, a potential energy diagram is often used. This diagram illustrates the energy changes that occur during a chemical reaction. The reactants are located at the starting point of the diagram, while the products are located at the ending point. The difference in energy between the reactants and products represents the overall energy change of the reaction, and the activated complex represents the highest energy point or transition state.
By analyzing the potential energy diagram, scientists can determine the activation energy by measuring the vertical distance between the reactants and the activated complex. This information can then be used to calculate the reaction rate using the rate constant and the concentration of reactants.
In conclusion, determining the activation energy and reaction rates is essential for understanding the speed and efficiency of chemical reactions. It allows scientists to optimize reaction conditions and develop new catalysts that can lower the activation energy and increase the reaction rate. By studying the potential energy diagram and using mathematical equations like the Arrhenius equation, scientists can gain valuable insights into the energetic and kinetic aspects of chemical reactions.
Examining Exothermic and Endothermic Reactions
Exothermic and endothermic reactions are two types of chemical reactions that involve the release or absorption of energy, respectively. Understanding these reactions and their potential energy diagrams is crucial in studying the behavior and properties of different substances.
An exothermic reaction is a chemical reaction that releases energy in the form of heat or light. It is usually accompanied by a decrease in the potential energy of the system. In an exothermic reaction, the reactants have a higher potential energy than the products, and the difference in energy is released as heat. The potential energy diagram of an exothermic reaction typically shows a downhill slope, indicating the decrease in energy.
On the other hand, an endothermic reaction is a chemical reaction that absorbs energy from the surroundings. It is usually accompanied by an increase in the potential energy of the system. In an endothermic reaction, the reactants have a lower potential energy than the products, and the difference in energy is absorbed from the surroundings. The potential energy diagram of an endothermic reaction typically shows an uphill slope, indicating the increase in energy.
Examining and analyzing potential energy diagrams can provide valuable insights into the nature of exothermic and endothermic reactions. By studying the changes in potential energy and the energy released or absorbed, scientists can better understand the dynamics and thermodynamics of chemical reactions. Additionally, potential energy diagrams can help predict the conditions under which a reaction is more favorable or the amount of energy required for a specific reaction.