
Stoichiometry is a fundamental concept in chemistry that involves quantifying the relationships between reactants and products in a chemical reaction. It allows scientists to determine the amount of each substance involved in a reaction and predict the outcome. However, navigating the complex calculations and equations involved in stoichiometry can be a daunting task.
Fortunately, stoichiometry maze answers provide a guide through this intricate web of chemical calculations. These answers provide step-by-step solutions to stoichiometry problems, helping students and researchers understand the principles behind chemical reactions. By following the maze of calculations, learners can develop a deeper understanding of the concepts and gain confidence in their problem-solving skills.
The stoichiometry maze answers delves into various aspects of chemical reactions, such as balancing equations, determining limiting reactants, and calculating reaction yields. With each solved problem, learners can unravel the puzzles of stoichiometry and begin to see the connections between the mass, moles, and ratios of substances involved in a reaction.
Unlocking the secrets of chemical reactions through stoichiometry maze answers not only improves academic performance but also provides a foundation for understanding real-world applications. From determining the composition of medications to optimizing industrial synthesis processes, stoichiometry plays a critical role in various fields. So, whether you’re a student struggling to grasp the concepts or a researcher seeking to unravel the mysteries of a reaction, stoichiometry maze answers can be your key to success.
What is stoichiometry?
Stoichiometry is a branch of chemistry that deals with the quantitative relationships between reactants and products in chemical reactions. It is a fundamental concept in chemistry and is used to determine the ideal ratio of reactants that will yield a desired amount of product. Stoichiometry is based on the law of conservation of mass, which states that matter cannot be created or destroyed in a chemical reaction.
In stoichiometry, chemical equations are balanced in order to ensure that the number of atoms of each element is the same on both sides of the equation. This allows chemists to determine the stoichiometric coefficients, which represent the relative amounts of reactants and products in a chemical reaction. These coefficients can be used to calculate the amount of reactants needed to produce a certain amount of product, or the amount of product that will be produced from a given amount of reactants.
Stoichiometry is essential in many areas of chemistry, including synthesis, analysis, and industrial processes. It allows chemists to make accurate predictions about the yield and efficiency of chemical reactions, and it provides a basis for understanding and quantifying the relationships between different chemical species. Stoichiometry calculations are also used in laboratory experiments to determine the purity, concentration, and identity of substances.
In summary, stoichiometry is the study of quantitative relationships in chemical reactions. It involves balancing chemical equations, determining stoichiometric coefficients, and using these coefficients to calculate the amount of reactants and products in a chemical reaction. Stoichiometry is an important tool in chemistry that enables scientists to predict and control the outcomes of chemical reactions.
Importance of stoichiometry in chemistry
Stoichiometry is a fundamental concept in chemistry that plays a crucial role in understanding and predicting chemical reactions. It allows chemists to determine the quantitative relationships between reactants and products in a chemical reaction, providing valuable information on the amount of substances involved.
Stoichiometry allows chemists to calculate the theoretical yields of products, which is important in both academic and industrial applications. These calculations help determine the efficiency of a reaction and assist in the design of chemical processes and synthesis of compounds. It also enables chemists to determine the limiting reactant, a key factor in predicting how much of a product can be obtained from a given amount of reactants.
The concept of stoichiometry is also crucial in balancing chemical equations. By following the principle of conservation of mass, chemists can ensure that the number of atoms on both sides of the equation remains equal. This fundamental skill is necessary for accurately representing chemical reactions and for solving complex problems in various fields, such as environmental science, pharmacology, and materials science.
Furthermore, stoichiometry plays a vital role in understanding the composition of substances and the stoichiometric relationships between different compounds. By analyzing the ratios of elements and compounds in a reaction, chemists can determine the empirical formula of a compound or identify unknown substances. This knowledge is essential in fields such as forensic chemistry and analytical chemistry, where the identification and characterization of compounds are critical.
In conclusion, stoichiometry is of utmost importance in chemistry due to its ability to provide quantitative information about chemical reactions, calculate theoretical yields, balance equations, and determine the composition of substances. It is a fundamental skill that enables chemists to understand the fundamental principles of chemistry and apply them in various scientific and industrial endeavors.
Understanding the stoichiometry maze
Stoichiometry is a fundamental concept in chemistry that deals with the quantitative relationships between reactants and products in chemical reactions. It allows us to calculate the amount of substances involved in a reaction and predict how much product will be formed.
To navigate through the stoichiometry maze, one must understand the concept of a balanced chemical equation. A balanced equation provides the stoichiometric coefficients, which indicate the ratio of reactants and products in the reaction. These coefficients are used to convert between different units of measurement, such as moles, grams, or liters.
When solving stoichiometry problems, it is important to identify the given information, such as the initial amount of reactant or the desired amount of product. Utilizing the stoichiometric coefficients, one can set up conversion factors and solve for the unknown quantity using dimensional analysis.
In order to successfully navigate the stoichiometry maze, it is crucial to have a solid understanding of the concept and its applications. Practice and repetition are key to mastering stoichiometry, as it involves multiple steps and calculations. Additionally, being familiar with common conversion factors and chemical formulas will aid in solving stoichiometry problems efficiently.
Overall, stoichiometry is an essential tool in chemistry that allows us to quantify chemical reactions and make predictions about the quantities of substances involved. By understanding the concept and practicing the necessary calculations, one can successfully navigate the stoichiometry maze and solve complex problems with ease.
Explanation of the stoichiometry maze

In the study of chemistry, stoichiometry plays a crucial role in understanding the relationship between different elements and compounds in a chemical reaction. Stoichiometry helps determine the quantities of reactants and products involved in a reaction, as well as the ratios in which they combine.
The stoichiometry maze is a tool designed to challenge students’ understanding of stoichiometry by presenting complex chemical equations and requiring them to solve for specific quantities or ratios. The maze consists of a series of interconnected paths, each representing a step in the stoichiometric calculations. Students must correctly navigate through the maze by solving each step in order to reach the final answer.
The maze typically starts with a balanced chemical equation, which provides the starting point for the stoichiometric calculations. Students must analyze the equation to determine the molar ratios between the different reactants and products. This information is then used to convert between moles of one substance to moles of another, using the concept of the mole ratio.
As students progress through the maze, they encounter different challenges, such as converting between moles and grams or determining the limiting reactant in a given reaction. These challenges require a solid understanding of stoichiometry principles and the ability to apply them in various scenarios.
The stoichiometry maze serves as a fun and interactive way for students to reinforce their understanding of stoichiometry and practice their problem-solving skills. By successfully navigating through the maze and reaching the correct answers, students gain confidence in their ability to apply stoichiometry principles to real-world chemical reactions.
Step-by-step guide to solve the stoichiometry maze
Stoichiometry is the branch of chemistry that deals with the quantitative relationships between reactants and products in chemical reactions. It allows us to calculate the amounts of substances needed or produced in a chemical reaction. Solving stoichiometry problems requires a systematic approach and following a few key steps can help navigate through the maze of equations and ratios.
Step 1: Write and balance the chemical equation
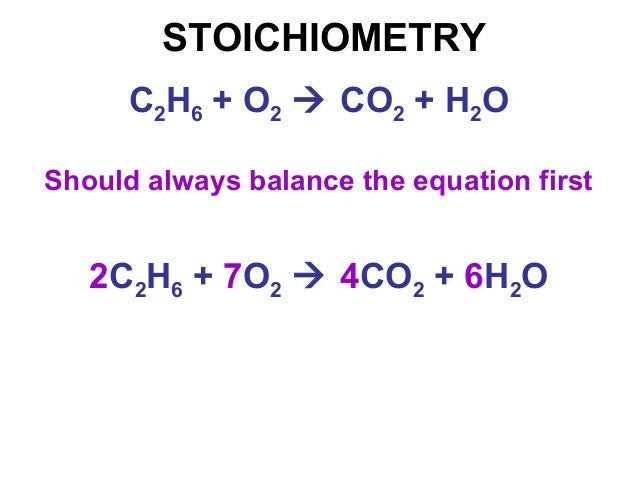
- Start by writing the chemical equation for the reaction, making sure it is properly balanced.
- If the equation is not balanced, adjust the coefficients in front of the molecules to ensure that the number of atoms on both sides of the equation is equal.
Step 2: Identify the known and unknown quantities
- Determine what quantities are given in the problem and what quantity you are trying to find.
- Write down the known quantities and label the unknown quantity as ‘x’.
Step 3: Convert known quantities to moles
- Use the molar mass of the given substance to convert the known quantity to moles.
- Use the formula moles = mass / molar mass to make the conversion.
Step 4: Use the balanced equation to set up ratios
- Examine the coefficients in the balanced equation and set up a ratio between the known and unknown substances.
- The ratio is based on the stoichiometric coefficients in the balanced equation.
Step 5: Use the ratio to find the unknown quantity
- Set up and solve a proportion using the known quantity in moles and the ratio determined in the previous step.
- Cross-multiply and solve for ‘x’, which represents the unknown quantity in moles.
Step 6: Convert moles to desired units
- Use the molar mass of the desired substance to convert the moles of the unknown quantity to the desired units (grams, liters, etc.).
- Use the formula mass = moles * molar mass to make the conversion.
Following these step-by-step instructions can help you solve stoichiometry problems and navigate through the maze of equations and ratios. Practice is key to gaining proficiency in stoichiometry, so don’t hesitate to try different problems and experiment with different scenarios.
Common Mistakes and How to Avoid Them
Stoichiometry can be a challenging topic for students to grasp, and it is common for them to make mistakes along the way. However, by being aware of these common errors and understanding how to avoid them, students can improve their understanding of stoichiometry and perform better on assessments.
1. Incorrectly identifying the given and unknown quantities:
One common mistake in stoichiometry problems is failing to correctly identify the given and unknown quantities. It is essential to carefully read the problem and identify the relevant information before starting any calculations. Often, the given quantities will be provided in the problem statement, while the unknown quantity will be what you are asked to find.
2. Using the wrong stoichiometric ratio:
Another frequent error is using the wrong stoichiometric ratio when converting between different substances. It is crucial to consult the balanced chemical equation and use the correct coefficients to establish the mole-to-mole ratio. These coefficients represent the number of moles of each substance involved in the reaction and are essential for accurate stoichiometric calculations.
3. Failing to account for limiting reagents:

One of the most significant mistakes in stoichiometry problems is not considering the limiting reagent. The limiting reagent is the reactant that is completely consumed in a chemical reaction, limiting the amount of product that can be formed. Failing to identify and account for the limiting reagent can lead to incorrect calculations and inaccurate results. It is important to determine which reactant is limiting and base calculations on its stoichiometry.
4. Inaccurate conversions between mass and moles:
Converting between mass and moles is a fundamental aspect of stoichiometry, and mistakes in these conversions can lead to inaccurate results. It is crucial to use the molar mass of a substance to determine the number of moles present. Students should double-check their conversions and make sure they are using the correct molar mass value for each substance involved in the calculation.
By being aware of these common mistakes and taking the time to understand the underlying concepts of stoichiometry, students can improve their problem-solving skills and successfully navigate the challenges of this topic. Regular practice and reviewing the steps involved in stoichiometric calculations can also help reinforce learning and minimize errors.
Benefits of practicing stoichiometry maze

Stoichiometry is a fundamental concept in chemistry that involves the calculation of quantities in chemical reactions. It is an essential skill for any student studying chemistry, as it provides a quantitative understanding of chemical reactions and allows for the prediction of reaction outcomes. One effective way to practice stoichiometry is through the use of stoichiometry mazes.
Engages students in problem-solving: Stoichiometry mazes present students with a series of interconnected questions that require them to apply their knowledge of stoichiometry to navigate through the maze. By solving these problems, students develop critical thinking and problem-solving skills, as they need to analyze the given information, determine the appropriate calculations to use, and apply their understanding of stoichiometry to find the correct path.
Reinforces understanding of stoichiometry concepts: Stoichiometry mazes cover a range of stoichiometry topics, including balancing chemical equations, calculating molar ratios, determining limiting reactants, and calculating yields. By practicing stoichiometry through mazes, students reinforce their understanding of these concepts and improve their ability to apply them in various scenarios.
Provides immediate feedback: Stoichiometry mazes typically come with answer keys, allowing students to check their answers immediately after completing the maze. This immediate feedback is valuable as it helps students identify any mistakes or misconceptions they may have and enables them to correct them promptly. It also boosts students’ confidence and motivation, as they can see their progress and improvement as they navigate through more challenging mazes.
Offers an interactive and engaging learning experience: Stoichiometry mazes add an element of fun and challenge to the learning process. They often feature colorful and visually appealing designs that capture students’ attention and make the practice more enjoyable. The interactive nature of mazes makes learning stoichiometry more engaging and entertaining, promoting active participation and deeper understanding of the subject.
In conclusion, practicing stoichiometry through mazes offers several benefits for students studying chemistry. It engages them in problem-solving, reinforces their understanding of stoichiometry concepts, provides immediate feedback, and offers an interactive and engaging learning experience. By incorporating stoichiometry mazes into their study routine, students can enhance their proficiency in stoichiometry and build a solid foundation for further studies in chemistry.